
Introduction
Every medical product business knows the scenario: your 3PL is dropping the ball with missed shipments, inventory discrepancies, and poor compliance handling, but switching feels even riskier than staying. Switching means keeping orders flowing while your entire logistics infrastructure changes underneath them. One misstep and you're looking at lost orders, compliance violations, or both.
3PL migration means transferring your inventory, order fulfillment operations, and system integrations from your current logistics provider to a new one. According to a 2024 Gartner survey, 76% of logistics transformations fail to meet critical budget, timeline, or KPI metrics. For medical product businesses, the stakes climb higher—regulatory compliance can't slip during the transition.
This guide covers exactly how to execute that transition without losing orders, missing compliance requirements, or inheriting the same problems you're trying to leave behind.
TL;DR
- A successful 3PL switch follows four phases: clean exit, parallel onboarding, strategic inventory move, and post-go-live monitoring.
- The biggest risk is a poorly timed cut-off date or untested system integration—both have clear fixes if you plan ahead.
- Medical product businesses must verify that the new 3PL meets FDA, ISO, and GMP compliance requirements before moving inventory.
- Migrating inventory in phases keeps fulfillment running while the switch happens.
- Skipping the pre-move inventory audit is the most common cause of post-migration disputes and missing stock.
When Is It Time to Switch Your 3PL Provider?
Some warning signs are hard to ignore. A 3PL relationship becomes a liability when you're regularly dealing with:
- Persistent shipping errors or incorrect orders
- Inventory discrepancies that don't reconcile
- Little to no visibility into reporting or tracking
- Poor communication from your logistics team
- An inability to scale with your order volume
For medical product businesses, compliance failures carry even higher stakes. Improper storage of temperature-sensitive items or mishandled regulatory documentation can expose your business to legal and financial risk—not just operational headaches.
Not every switch is reactive. Some businesses simply outgrow their 3PL — order volumes climb, new sales channels open up (Amazon, international), or compliance requirements become more complex than a generalist provider can handle. Staying with the wrong partner through a high-demand period creates more risk than a well-planned migration ever would.
What to Prepare Before Starting Your 3PL Migration
The quality of your preparation directly determines how smooth the migration will be. Before any conversations with a new 3PL begin, pull and review your current contract. Identify:
- The notice period required to terminate (typically 30–90 days)
- Any exit or inventory removal fees
- Your rights to a complete data export of all order history and inventory records
Equipment and Data Requirements
Compile a Master Product File covering every SKU:
- Product name, UPC/barcode, dimensions, weight, declared value
- Special handling requirements
- For medical products: lot numbers, expiration dates, serialization data, hazmat classifications, and any regulatory tracking data
This file becomes the foundation for onboarding your new 3PL's warehouse management system (WMS). Poor master data configuration causes misrouted orders, incorrect packaging, and inventory discrepancies from day one.
Compliance and Handling Readiness
Identify any regulatory or handling requirements your products demand during transit and storage:
- FDA compliance for medical devices
- ISO and GMP standards
- Cold chain requirements
- HIPAA-safe data handling
Confirm these requirements in writing with any prospective new 3PL before committing. Compliance gaps discovered mid-migration are costly and hard to reverse — ask for written verification of certifications and compliance protocols before you sign anything.

How to Migrate from One 3PL to Another Without Losing Orders
Step 1: Exit Your Current 3PL With a Clear Plan
Formally submit your termination notice in writing as soon as you have identified your new provider, so the notice period clock starts immediately. Communicate professionally—your current 3PL still controls your inventory during the wind-down and you need their cooperation.
Conduct a full physical inventory audit at the current facility before any product moves. Document:
- Every SKU and quantity
- Lot numbers and expiration dates
- Any damage or discrepancies
For medical products, this audit is your chain-of-custody baseline. Without it, there's no reliable way to verify what arrived at the new facility versus what was lost or damaged in transit.
Set a firm cut-off date: the last day your current 3PL will fulfill and ship customer orders. This date must be coordinated precisely with your new 3PL's go-live date to close any fulfillment gap. Build in a buffer for in-transit orders placed before the cut-off to be completed by the old provider.
Step 2: Onboard Your New 3PL in Parallel
While the wind-down is running, begin onboarding your new 3PL as a parallel workstream. A quality new partner will assign a dedicated onboarding contact and provide a documented project plan—if they do not, that's a red flag.
For medical product businesses specifically, verify that the new facility meets:
- FDA-cleared packaging and storage requirements
- ISO and GMP standards
- Proper infrastructure to handle sensitive or regulated items
Provide your Master Product File to the new 3PL so they can configure their WMS with accurate product data before any inventory arrives. Confirm that all product attributes—dimensions, weight, special handling flags, lot tracking fields—have been entered correctly.
Connect your eCommerce stores (Shopify, WooCommerce, Amazon Seller Central, etc.) to the new 3PL's WMS and run test orders before go-live. Process several dummy transactions to verify:
- Orders flow in correctly
- Inventory levels update on your storefront
- Tracking numbers are generated and sent back to your sales channels
As DC Velocity notes, "interfaces are easy, integrations are hard." Do not go live until this integration test passes without errors.
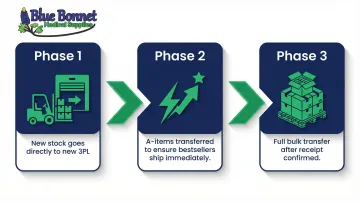
Step 3: Execute the Physical Inventory Move With a Phased Strategy
Use a phased migration rather than moving everything at once:
- Route your next inbound shipment from your manufacturer directly to the new 3PL — they start receiving and storing your product before the bulk transfer happens
- Move your highest-velocity SKUs (A-items) first so your bestsellers are available to ship from the new facility right away
- Transfer remaining stock once the new facility has confirmed accurate receipt of priority items
Create a detailed Advance Ship Notice (ASN) for every inbound shipment—a digital packing list specifying exactly which SKUs and quantities are in each delivery. This allows the new facility to receive your stock quickly and accurately.
It also creates a verifiable record to cross-reference against your pre-move inventory audit. For medical products, the ASN should include lot numbers and any required regulatory tracking data.
Coordinate the freight using LTL or FTL carriers. Check whether your new 3PL has established carrier relationships that can reduce transfer costs. Confirm the carrier is approved for your product type — this matters most for medical devices with temperature or specialized handling requirements.
Step 4: Go Live and Monitor the First Wave of Orders
On go-live day, switch your order routing from the old 3PL to the new one. Communicate the cut-over clearly and in writing to both providers so there is no ambiguity about which facility is responsible for fulfilling orders at any given moment.
Monitor the first 24–48 hours of live fulfillment closely:
- Track the first batches of orders in real time
- Verify that pick-and-pack accuracy matches expectations
- Confirm that tracking numbers are being pushed correctly to your sales channels and to customers
- Identify and escalate any discrepancies immediately while your onboarding contact is still actively engaged
After the first week of stable operation, run a post-go-live review. Compare your opening inventory count at the new facility against the pre-move audit baseline and reconcile any discrepancies. Then set the KPIs you'll use to measure ongoing performance:
- Order accuracy rate
- Fulfillment cycle time
- Return processing time
Factors That Make or Break a 3PL Migration
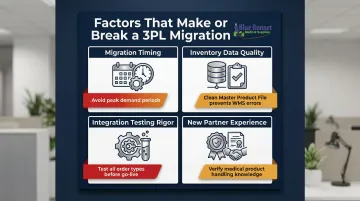
Two businesses can follow the same migration steps and achieve very different results—because several variables outside the steps themselves determine whether the transition is smooth or chaotic. Four variables consistently separate smooth transitions from costly ones:
Timing relative to peak demand: Migrating during high-volume periods (Q4, a major launch, a promo campaign) adds serious risk. In 2019, Asos initiated a warehouse technology transition just before peak season—the system failed to register inventory and returns, took months to resolve, and cost the company $30 million. Schedule your migration during a low-demand quarter instead.
Quality of inventory data going in: Errors in your Master Product File—outdated SKUs, missing attributes, incomplete records—multiply inside the new WMS. Clean, validated data prevents misrouted orders, incorrect packaging, and inventory discrepancies from the first day of operation.
Integration testing rigor: Rushing or skipping the dummy order test phase is one of the most common causes of stalled orders at go-live. Tests should cover standard single-item orders, multi-SKU orders, special handling requirements, and return orders.
New partner's experience with your product type: A generalist 3PL may not understand the handling, documentation, or compliance requirements specific to medical products. Before committing, verify the new partner has processed similar products and can demonstrate familiarity with applicable regulations.
Common Mistakes That Lead to Lost Orders During a 3PL Transition
No pre-move inventory audit: Without a verified baseline count before any product moves, you have no way to confirm everything arrived at the new facility. Disputes become impossible to resolve, and lost stock goes undetected until customer orders start failing.
Misaligned cut-off and go-live dates: When the old 3PL stops fulfilling before the new one is ready, orders queue up with no one processing them — the most common cause of visible delays during transitions. Both dates must be coordinated to the day, with a clear plan for orders placed in between.
Going live before the integration is tested: Routing real orders through an unvalidated WMS invites a cascade of first-wave errors. Wrong items get shipped, tracking numbers stop updating, and inventory levels desync across your sales channels. Test against your actual order types before flipping the switch.
Each of these mistakes is preventable with the right transition plan in place.
Frequently Asked Questions
How long does it typically take to migrate from one 3PL to another?
A well-planned migration typically takes 30 to 90 days from initiating the new 3PL onboarding to going live, depending on inventory volume, contract notice periods, and system integration complexity.
What happens to orders placed during the transition period?
Orders placed before the cut-off date are fulfilled by the old 3PL; orders after go-live are handled by the new one. A clear cut-off date communicated to both providers prevents orders from falling through the gap.
Do I need to notify customers when switching 3PL providers?
Most customers don't need to know the back-end logistics are changing. If delays are expected during the transition window, a heads-up about the timeline protects customer satisfaction and cuts down on support tickets.
What is an Advance Ship Notice (ASN) and why does it matter?
An ASN is a digital packing list sent to the new 3PL before inventory arrives, listing every SKU and quantity. It enables accurate receiving and creates a verifiable record to cross-reference against your pre-move inventory audit.
How do I avoid losing inventory during a 3PL transition?
Conduct a full physical inventory audit at the old facility before any product moves, and cross-reference it against the ASN when inventory arrives at the new 3PL. This is the most reliable way to catch and resolve discrepancies before they impact orders.
What should medical product businesses specifically look for in a new 3PL?
Look for verified FDA, ISO, and GMP compliance, plus storage infrastructure built for sensitive or regulated items. Experience with chain-of-custody documentation is equally important — not all 3PLs understand the paper trail medical products require.


