
According to Texas Appeals Court rulings, vague contractual language like "best efforts" is legally unenforceable without objective numeric guidelines. For medical product businesses, this isn't just a customer service issue—it's a regulatory and financial liability.
This article explains what a fulfillment SLA actually is, breaks down every clause that belongs in one, and gives medical product businesses the specific benchmarks worth demanding before signing anything.
TLDR:
- A fulfillment SLA defines services, timelines, accuracy targets, and accountability between you and your 3PL
- Complete SLAs cover receiving, outbound processing, order accuracy, returns, reporting, and escalation—not just shipping speed
- Medical businesses need FDA handling compliance, temperature/storage standards, and regulatory documentation clauses
- Demand: receiving turnaround within 1–2 business days, 99%+ order accuracy, defined cutoff times with clock rules
- Red flags: vague "shipped" definitions, no penalties for breaches, missing compliance clauses
What Is a Fulfillment SLA (and Why Medical Businesses Can't Afford Vague Contracts)
A fulfillment SLA is a formal, written agreement between a business and its 3PL provider that specifies which services will be delivered, how performance will be measured, what timelines apply, and what happens when commitments are not met. Unlike general service contracts, SLAs are metric-driven and clock-bound. They establish objective standards that can actually be enforced—and in medical logistics, that distinction matters more than most businesses realize.
The "Shipped" Problem
The word "shipped" alone creates costly disputes. Does it mean:
- Label printed?
- Order completed and staged?
- Carrier accepted?
A strong SLA defines two distinct milestones: warehouse completion timestamp (when the order is packed and ready) and carrier acceptance timestamp (when the carrier physically takes possession). Tracking the gap between these events reveals missed pickups and dock congestion problems that a label-printed metric will never surface.
The Medical Product Stakes
That gap between "printed" and "picked up" isn't just a logistics nuance—for medical product businesses, it can trigger account-level consequences. Amazon enforces strict Order Performance policies: Order Defect Rate must stay under 1%, Late Shipment Rate under 4%, and Pre-Fulfillment Cancellation Rate under 2.5%. Exceeding these triggers listing suppression or account deactivation.
Late or mis-shipped medical supplies also create:
- Failed customs clearances due to missing documentation
- Regulatory violations when temperature-controlled items aren't handled properly
- Patient-level consequences when medical devices don't arrive on time
When the product is a medical device or supply, a vague SLA becomes a compliance gap—one with consequences that go well beyond a refund request.
SLAs Cover the Entire Pipeline
An SLA applies to the complete fulfillment pipeline, not just outbound shipping:
- Receiving velocity: How quickly inventory moves from dock to available-to-pick status
- Inventory accuracy: Alignment between system records and physical stock
- Returns processing: Turnaround time from receipt to restocking, repair, or disposal
- Compliance documentation: Proper handling records for FDA, ISO, and GMP requirements
All of these directly affect outbound performance. If inventory sits unprocessed on the dock, it can't be picked—making receiving SLAs as critical as shipping cutoffs.
What Should Be Included in a Fulfillment SLA
Service Description and Scope
Start by listing every service covered, with enough specificity that a new employee could use it as an onboarding checklist:
- Receiving and put-away
- Pick and pack operations
- Outbound shipping
- Returns processing
- Value-added services (kitting, labeling, special packaging)
Equally important: explicitly state what is excluded. Without clear boundaries, scope disputes arise over services you assumed were standard.
For example, Bluebonnet Medical Supplies separates standard operations (FDA-compliant receiving, storage, returns processing) from custom arrangements (product restoration, special labeling) in writing — so both sides know exactly what the base contract covers before work begins.
Performance Metrics
Vague language like "accurate" or "on-time" is unenforceable. Tie every metric to a number:
| Metric | Definition | Best-in-Class Target |
|---|---|---|
| Order Accuracy Rate | Percentage of orders fulfilled without errors | ≥99.68% |
| On-Time Shipment Rate | Orders shipped at planned time (off dock, in transit) | ≥99.5% |
| Inventory Accuracy | Alignment between system records and physical stock | ≥99.6% |
| Return Processing Time | Initial receipt to final disposition | ≤14 days |

Benchmarks based on WERC 2025 DC Measures
If a 3PL resists putting numbers on paper, that resistance itself tells you something.
Cutoff Times and Clock Rules
Cutoff times must define:
- Clock start: When does processing time begin? (Order received vs. order confirmed in WMS)
- Clock stop: When does it end? (Label printed vs. carrier acceptance)
- Pause conditions: Which order states or exceptions pause the clock?
- Carrier pickup windows: How are carrier arrival delays factored in?
Ambiguity in clock rules turns every peak-season miss into a dispute. A reasonable D2C cutoff is orders placed by noon for same-day processing. The SLA must also specify what happens to orders placed after cutoff, on weekends, and during peak seasons.
Research shows that missing a predefined truck departure time by mere minutes can result in a full-day delay to the customer. Precise cutoff definitions prevent these disputes.
Responsibilities of Each Party
An SLA that only lists what the 3PL will do isn't a true agreement — it's a promise. Make it bilateral:
3PL Responsibilities:
- Timely processing within agreed windows
- Accurate inventory records
- Compliance documentation
- Issue notification and escalation
Client Responsibilities:
- Accurate order information
- Proper inbound shipping notices (ASNs)
- Timely payment
- Reasonable forecast accuracy
Symmetric responsibilities also give you standing to dispute SLA credits when a 3PL can demonstrate the failure originated on your side.
Issue Resolution and Escalation Procedures
SLAs must include a structured escalation path:
- Who to contact first for different issue types
- Response time commitments by issue severity
- How service failures are documented
- What remedies apply when targets are missed
Many 3PL SLAs adopt the P1/P2/P3/P4 severity framework from ITIL service management:
| Priority | Example | Response Target | Resolution Target |
|---|---|---|---|
| P1 - Critical | Total WMS outage; facility halted | 15 minutes | 4 hours |
| P2 - High | EDI failure; workaround exists | 30 minutes - 1 hour | 8 hours |
| P3 - Medium | Single station down; minor impact | 2-4 hours | 3 business days |
| P4 - Low | Cosmetic issue; general inquiry | 1 business day | 5 business days |

Without defined escalation paths, critical issues get lost in general support queues.
Penalties, Credits, and Incentives
Consequences are what separate an enforceable SLA from a wishlist. Look for three components:
Service Level Credits (percentage of monthly fees):
- 99.75% to 99.94% performance: 5% credit
- 99.50% to 99.74% performance: 7.5% credit
- Below 99.50% performance: 10% credit
Corrective Action Plans (CAPs): If a 3PL repeatedly breaches SLAs, the contract should mandate a CAP to identify root causes and remedy deficiencies.
Incentives: Some agreements include "earn-back" clauses allowing 3PLs to recover credits if they sustain high performance over subsequent periods.
If the SLA has no penalty clause, a missed target carries no formal consequence. The 3PL has every incentive to prioritize clients whose contracts do bite.
What to Demand: Minimum Standards Worth Fighting For
A 3PL presenting an SLA with no defined benchmarks is presenting a blank check. These are the specific minimums worth holding the line on:
Outbound Order Accuracy: 99% or Above
Industry best-in-class is 99.68%. Anything below 99% means one in every 100 orders contains an error—unacceptable when Amazon's Order Defect Rate threshold is 1%.
Receiving Turnaround: 1–2 Business Days
Best-in-class dock-to-stock cycle time is under 3.5 hours. Demand that receiving SLAs measure time from dock arrival to inventory stowed in pick locations, not just "received."
Defined Cutoff Times with Clock Rules
"Same-day" without a cutoff definition is not an SLA. Demand written cutoff times (e.g., orders by noon ship same day) with explicit clock-start and clock-stop rules.
Two-Milestone Outbound Definition
Track both warehouse completion timestamp and carrier acceptance timestamp. The gap between these events reveals missed pickups and dock congestion that label-printed metrics hide.
Real-Time or Near-Real-Time Reporting
A client portal with daily order status, KPI dashboards, and historical transaction logs is a baseline requirement. If a 3PL cannot show inventory and order progress in real time, SLA measurement depends entirely on their self-reporting—a built-in conflict of interest.
Defined Exception Policy
Ask the 3PL to specify in writing which conditions pause the fulfillment clock:
- Damage on receipt
- Missing documentation
- Carrier delays
- Force majeure events
Also specify which conditions do NOT pause the clock. An unscoped exception policy allows any miss to be reclassified as excused.
Receiving SLAs and Outbound SLAs: Two Clocks, One Pipeline
Receiving is the hidden driver of every outbound metric. If inventory sits on the dock unprocessed, it cannot be picked—making on-time delivery impossible regardless of outbound performance.
Receiving SLA Benchmarks
A receiving SLA measures time from dock arrival to inventory stowed in pick locations. Best-in-class benchmark: under 3.5 hours.
Receiving SLAs should handle exceptions explicitly:
- Damaged product
- Missing Advance Ship Notices (ASNs)
- Inventory discrepancies
Each of these can pause the receiving clock — but the SLA should define every pause condition explicitly and set a hard maximum total receiving window, even when exceptions apply.
Strong receiving performance creates the foundation, but outbound SLAs determine whether that inventory reaches customers on time and in compliance.
Segment Outbound SLAs by Channel
D2C parcel orders, B2B retail purchase orders, and international shipments each carry different compliance milestones — and different consequences for missing them. A single outbound SLA that averages all channels together obscures where failures actually happen.
Channel-specific considerations:
- D2C: Requires same-day order cutoffs, parcel carrier pickup windows, and Amazon seller performance thresholds
- B2B: Demands retailer routing compliance, correct pallet configurations, and confirmed delivery appointments
- International: Involves customs documentation deadlines, carrier booking cutoffs, and country-specific regulatory requirements

Compliance SLA Clauses: What Medical Product Businesses Must Require
Medical product businesses face regulatory requirements that general-merchandise SLAs never address. Effective February 2, 2026, the FDA's Quality Management System Regulation (QMSR) incorporates ISO 13485:2016 by reference, requiring documented procedures for preserving product conformity during storage, handling, and distribution.
FDA Handling Compliance
A fulfillment partner handling medical supplies must demonstrate:
- Verified FDA-compliant packaging and handling practices
- GMP-aligned storage conditions covering temperature control and contamination prevention
- HIPAA-safe data handling for any fulfillment involving Protected Health Information
These must appear as verifiable SLA commitments, not marketing claims. Ask for documented certifications (FDA clearance, ISO, GMP) and request to see how those standards are operationalized in the warehouse.
Bluebonnet Medical Supplies holds FDA-cleared medical packaging services alongside ISO and GMP compliance — ask any 3PL candidate to show the same documentation before signing.

HIPAA Applicability
The HIPAA Privacy Rule requires Business Associate Agreements (BAAs) with any vendor that handles Protected Health Information (PHI). Whether your fulfillment data triggers BAA requirements depends on what it contains:
- Conduit exemption applies to USPS and private couriers that simply transport packages
- BAA required when a 3PL stores or processes data containing PHI — patient names, addresses, or specific device identifiers all qualify
- Verify before signing that your 3PL will execute a BAA if your fulfillment data touches any PHI
International Documentation Compliance
FDA and HIPAA obligations apply domestically — cross-border shipments layer on a separate set of documentation requirements:
- U.S. Customs: Entry summary (CBP Form 7501), Harmonized Tariff Schedule (HTS) codes, FDA product codes, Affirmations of Compliance
- EU MDR: CE marking verification, proper labeling, Eudamed registration, storage/transport condition documentation
Missing Device Registration or Listing numbers cause automated FDA PREDICT system failures, delaying entry. The SLA should define the 3PL's responsibility for preparing and submitting this documentation accurately so products are not held at customs.
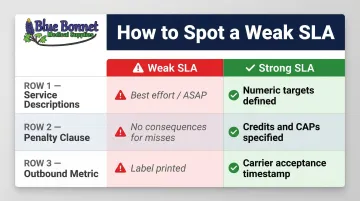
How to Spot a Weak SLA Before You Sign
Three Common Weak SLA Patterns
- Vague service descriptions — phrases like "best effort" or "as soon as possible" carry no numeric targets and are legally unenforceable
- No penalty clause — without consequences for missing targets, the SLA is aspirational, not binding
- Label-printed metrics — measuring "label printed" instead of "carrier accepted" lets a 3PL claim compliance while orders sit on a dock
The Pre-Signature Test
Ask the 3PL to walk you through a live day in their client portal:
- Trace one order from intake through carrier acceptance
- Show you one example of an exception being handled
- Display current KPI dashboards
If they cannot do this, SLA enforcement will depend on your ability to identify problems yourself and argue about them.
Scalability Language
Once you've confirmed a 3PL can demonstrate day-to-day performance, push them on high-volume scenarios. Find out whether SLA targets are maintained during peak periods — holiday season, product launches — and whether the contract includes volume buffers or requires renegotiation when order volume grows significantly.
Without scalability provisions, a 3PL that meets SLAs at 1,000 orders/month may fail completely at 5,000 orders/month.
Frequently Asked Questions
What is a fulfillment SLA?
A fulfillment SLA is a formal agreement between a business and its 3PL defining specific services, measurable performance targets (accuracy, speed, receiving time), timelines, and consequences for not meeting commitments. It differs from general contracts by being metric-driven and time-bound.
What should be included in a fulfillment SLA?
Core components include:
- Service scope and performance metrics (order accuracy, on-time shipment, inventory accuracy)
- Cutoff and clock rules, plus each party's responsibilities
- Issue escalation procedures and reporting frequency
- Penalty or credit terms for missed or exceeded targets
What is the primary goal of order fulfillment?
The primary goal is to get the right product to the right destination accurately and on time. For medical product businesses, this also includes ensuring regulatory compliance (FDA, GMP, HIPAA) throughout the handling and shipping process.
What are P1, P2, P3 and P4 ticket SLAs?
These are issue severity tiers in SLA escalation frameworks. P1 is critical (complete service failure), P2 is high (significant disruption), P3 is medium (non-critical issue), and P4 is low (minor request). Each tier carries a defined response time commitment.
What happens if a 3PL misses its SLA targets?
Consequences depend on what's written in the SLA. Well-structured agreements include service credits (percentage of monthly fees), required corrective action plans, or expedited shipping at the 3PL's cost. If the SLA has no penalty clause, a missed target has no formal consequence.
Do medical product companies need special SLA terms?
Yes. Standard SLA templates don't cover FDA handling compliance, GMP storage requirements, HIPAA-safe data practices, or international documentation for regulated products. Medical businesses must explicitly negotiate these clauses—including documented certifications and verified warehouse procedures—into any 3PL agreement.


