
Introduction
Medical product businesses reach a breaking point when logistics outgrow in-house capabilities. Choosing a 3PL partner is a compliance and safety decision as much as a cost one. The wrong partner can mean improper storage, customs holds, regulatory violations, or damaged products that erode customer trust.
The stakes are particularly high for regulated medical items. FDA warning letters issued in 2024–2025 show that distribution failures—improper storage, labeling errors, and inadequate documentation—continue to trigger enforcement actions against medical device companies. For businesses shipping internationally or selling through Amazon, the compliance bar rises even higher.
That complexity is driving demand: the global medical device 3PL market is projected to reach $47.01 billion by 2030, growing at 9.0% annually. And when companies do find the right fit, the results show — 89% of shippers report successful 3PL relationships, with 82% crediting improved customer service. This checklist walks you through exactly what to evaluate before signing with a partner.
TL;DR
- Medical 3PLs must meet FDA, ISO, GMP, and HIPAA requirements — general fulfillment centers won't cut it
- Specialized storage, verified certifications, and strict handling protocols are non-negotiable for medical products
- Six factors matter most: compliance, storage conditions, inventory tech, scalability, shipping network, and pricing transparency
- Walk away from any 3PL with vague compliance claims, no dedicated account contact, or opaque fee structures
- The right partner protects your products, keeps you compliant, and gives you room to grow
What Is a 3PL Fulfillment Partner?
A third-party logistics (3PL) provider manages warehousing, order fulfillment, inventory management, and shipping on behalf of another business. This allows companies to focus on growth and customer relationships instead of logistics operations.
For medical product companies, that outsourcing relationship carries extra weight. You retain control of your brand, sales strategy, and customer relationships, but the 3PL executes the physical fulfillment operations. Any compliance failures in that execution become your liability — which means choosing the right partner matters more than it might in other industries.
Core services a full-service 3PL provides:
- Receiving and storage of inventory
- Pick-and-pack order fulfillment
- Domestic and international shipping coordination
- Returns processing and inspection
- Real-time inventory tracking and reporting
Why Medical Product Businesses Need a Specialized 3PL
Medical products—devices, supplies, diagnostic equipment—carry unique handling, storage, and documentation requirements that general fulfillment warehouses aren't equipped to meet. Temperature sensitivity, sterile packaging protocols, and serialization tracking are only part of the picture—regulatory compliance is the other half.
The Compliance Dimension
Shipping medical products without FDA-cleared processes, GMP-compliant handling, or HIPAA-safe data practices creates serious risks:
- Customs delays and product rejections at international borders
- Regulatory penalties and warning letters from the FDA
- Product liability exposure from damaged or contaminated goods
- Loss of Amazon selling privileges for non-compliant FBA shipments
FDA enforcement is active. Recent warning letters cite distribution failures including inadequate storage controls, missing UDI labeling in Device History Records, and failure to establish documented packaging requirements.
These violations don't just affect manufacturers—they extend to 3PLs acting as initial distributors or packagers.
Amazon and International Requirements
For businesses selling through Amazon or shipping internationally, the compliance bar is even higher. Amazon prohibits FBA for prescription and professional-use medical devices—these must be fulfilled via Merchant Fulfillment Network (MFN) by a compliant 3PL.
International medical product exports require FDA export certificates, proper HTS classification, and country-specific import documentation. A 3PL that understands cross-border medical regulations prevents costly shipment holds and rejections.
The 3PL Selection Checklist: 6 Factors That Matter Most
Each factor below maps directly to an operational risk or business outcome—not just a "nice to have." Use this as a vetting framework before signing any contract.
Regulatory Compliance & Certifications
Verify the 3PL holds relevant certifications for medical product handling:
- FDA-cleared packaging and handling processes
- ISO 13485:2016 compliance for medical device quality management
- GMP (Good Manufacturing Practice) compliance
- HIPAA-safe data handling practices

Ask for documentation, not just verbal assurances. A compliant 3PL will have no hesitation providing proof of active certifications. The FDA's Quality Management System Regulation (QMSR), effective February 2, 2026, incorporates ISO 13485:2016 by reference, making ISO compliance a regulatory requirement for entities performing packaging, labeling, and initial distribution.
Confirm international shipping compliance: Can they ensure medical products clear customs without holds due to improper documentation or non-compliant packaging? Ask about their experience with FDA export certificates and country-specific medical import restrictions.
Specialized Storage & Handling Capabilities
Assess whether the warehouse is purpose-built or adapted for sensitive medical items:
- Proper shelving and climate control where needed
- Segregation of product types to prevent cross-contamination
- Protocols to prevent damage during storage and handling
- Staff training on medical packaging requirements
Ask specifically about pick-and-pack procedures: Are staff trained to handle medical packaging carefully? Are there quality checks before shipment? For medical devices, even minor packaging errors create liability issues.
Technology & Inventory Visibility
Real-time inventory management is non-negotiable. Your 3PL should integrate directly with your sales channels—Amazon, Shopify, your own website—so you always know stock levels, order status, and shipment tracking without manual effort.
Look for data reporting capabilities:
- Order accuracy rates
- Fulfillment speed metrics
- Inventory discrepancy reports
Top-performing warehouses achieve order-picking accuracy of ≥99.85%, according to the Warehousing Education and Research Council. For medical businesses, accurate inventory data is also a compliance requirement, not just a convenience.
Scalability & Flexibility
Your 3PL should scale with your business—handling volume spikes during product launches or seasonal demand without compromising accuracy or turnaround times. Look for partners who adapt to your workflow rather than forcing you into their template.
Signs of a genuinely flexible 3PL:
- Customizable storage configurations for your product types
- Adaptable labeling and shipping protocols per SKU or destination
- No rigid minimum volume requirements that penalize early growth
- Willingness to discuss your roadmap, not just your current order volume
Shipping Network & International Reach
**Confirm the 3PL has established carrier relationships** that cover your key markets—both domestic and international—and ask whether they offer negotiated carrier rates that pass savings to you. This reduces per-order shipping costs at scale, and the savings compound quickly on high-volume accounts.
For businesses exporting medical products, verify the 3PL's experience with:
- International documentation requirements
- Customs compliance for medical goods
- Country-specific import restrictions that could affect your shipping lanes
Cost Structure & Transparency
Request a full breakdown of all fees:
- Receiving and storage (per pallet or per unit)
- Pick-and-pack charges
- Shipping costs
- Returns processing
- Any minimum volume commitments or surcharges
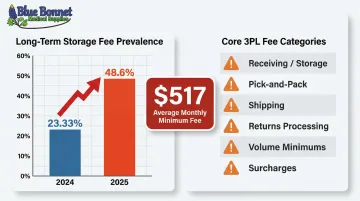
Hidden fees are one of the most common pain points businesses report after signing with a 3PL. In 2025, 48.6% of warehouses charged long-term storage fees (up from 23.33% in 2024), and average monthly minimums rose to $517.
Evaluate total cost of partnership, not just line-item rates. A 3PL with carrier discounts, low error rates, and strong compliance may cost more upfront but saves significantly on chargebacks, reshipping, and regulatory penalties over time.
How Bluebonnet Medical Supplies Can Help
Bluebonnet Medical Supplies is a personal-approach 3PL built specifically for medical product businesses. Unlike large-volume 3PLs that slot every client into the same workflow, Bluebonnet handles storage, packing, and shipping around your specific compliance requirements.
Key differentiators:
- Storage protocols built for sensitive medical items, not repurposed general warehouse space
- FDA-cleared packaging with ISO, GMP, and HIPAA-safe handling built into daily operations
- Discounted shipping rates through direct carrier relationships, passed directly to clients
- Returns processing with product testing and restoration to recover value from returned inventory
- Flexible setup for Amazon sellers, direct-to-consumer sites, and international shipments

Bluebonnet's approach starts with understanding your specific needs rather than fitting you into a standardized system. Their team manages the compliance details—FDA documentation, proper labeling, customs clearance—so shipments clear without delays and products arrive intact.
If you're a medical product business looking for a compliant, personal-approach 3PL partner, reach out to Bluebonnet at info@bbmstx.com to discuss your specific fulfillment needs.
Conclusion
Choosing a 3PL fulfillment partner for a medical product business is a compliance decision as much as a logistics one. The right partner protects your products, your customers, and your business from costly operational and regulatory missteps.
Before you sign with anyone, run through these three steps:
- Use the 6-factor checklist as a non-negotiable starting point
- Ask every prospective 3PL for documentation of their certifications — not just verbal claims
- Revisit partner performance at least annually, or whenever your business scales
Your logistics needs will change as you grow. The right 3PL should be able to keep pace. At Bluebonnet Medical Supplies, that adaptability — alongside full FDA, ISO, and GMP compliance — is built into how we work with every client.
Frequently Asked Questions
How do I choose the right 3PL partner?
Match industry experience first, then verify compliance certifications relevant to your product type (FDA, ISO, GMP, HIPAA). Assess technology integration with your sales channels and evaluate cost transparency. For medical products, compliance credentials should be your first filter—without proper certifications, the partnership creates more risk than value.
How much does 3PL cost in the US?
3PL pricing typically includes storage fees ($20 per pallet/month), pick-and-pack fees ($3.25 per order), and shipping costs—with medical products adding premiums for climate control, serialization tracking, and compliance documentation. Pricing varies by volume, product type, and service level. Always request a full fee breakdown including receiving, returns processing, and monthly minimums before signing.
How to partner with a 3PL?
Define your logistics requirements first, then request quotes from vetted providers—ask for proof of certifications, not just claims. Negotiate contract terms covering fee structure, SLAs, and performance metrics. Then run integration testing and set clear onboarding timelines before going live.
What is the difference between a general 3PL and a medical 3PL?
A medical 3PL has certifications and facilities specifically designed for regulated products—FDA-cleared packaging, ISO 13485 compliance, GMP handling, and HIPAA-safe data practices. General 3PLs typically lack the storage protocols, trained staff, and documentation capabilities required for compliant handling, creating real regulatory and liability exposure.
Do I need an FDA-compliant 3PL for medical products?
Yes. Medical device and supply businesses are subject to FDA regulations that extend to packaging and distribution. Using a non-compliant 3PL can expose your business to regulatory violations, customs rejections, and product liability issues. The FDA's QMSR regulation applies to entities performing packaging, labeling, and initial distribution functions.
What certifications should a medical 3PL have?
Look for FDA-cleared packaging and handling processes, ISO 13485:2016 compliance for medical device quality management, GMP (Good Manufacturing Practice) compliance, and HIPAA-safe data handling where applicable. Always request proof of active certifications before signing a contract—compliant 3PLs will provide documentation without hesitation.


