
Introduction
A wrong 3PL partnership for medical products doesn't just mean delayed shipments. It can mean FDA warning letters from compliance violations, recalls from damaged regulated inventory, or shipments stuck at customs because documentation wasn't filed correctly.
The medical supply space operates under requirements that a generic 3PL isn't built for. Proper storage conditions, FDA-compliant packaging, GMP adherence, and HIPAA-safe handling aren't bonus features: they're baseline expectations that protect your business and your customers.
This guide covers the clearest red flags to spot before signing a contract, green flags that signal a trustworthy partner, and key questions to ask during a warehouse visit.
TLDR
- Medical 3PLs must hold verifiable compliance credentials (FDA registration, ISO 13485, GMP) — not just competitive rates
- Red flags: missing certifications, vague pricing, refused facility tours, zero medical industry references
- Legitimate partners respond quickly, share documented proof of compliance, and welcome scrutiny
- Tour the facility in person and verify how they handle temperature control, chain of custody, and recalls
- The right 3PL covers the full chain — storage, packing, domestic shipping, and international customs compliance
Why Evaluating a 3PL Matters More for Medical Products
Medical device recalls cost the industry up to $5 billion annually, with individual events running between $10 million and $50 million in direct costs. That exposure traces back, in many cases, to a logistics failure — not a product flaw.
Medical products face regulatory requirements that general consumer goods simply don't. FDA registration, ISO 13485 certification, GMP compliance, and HIPAA-safe handling are baseline expectations, not optional add-ons.
The compounding cost of a bad 3PL partnership for medical businesses is severe:
- Product integrity failures that trigger recalls and damage your brand reputation
- Customs holds on international shipments due to incomplete or incorrect documentation
- Loss of Amazon selling eligibility due to packaging violations or mislabeling
- Operational disruption from switching 3PLs mid-operation, which can take months and cost tens of thousands of dollars

That's why choosing the right 3PL isn't just a logistics decision — it's a compliance decision. The sections below break down exactly what to evaluate and which warning signs to watch for before you sign anything.
Red Flags to Watch Before Signing a 3PL Contract
The following red flags signal that a 3PL may not be equipped — operationally or legally — to handle medical products. Treat each one as a serious warning.
No Certifications or Unverifiable Compliance Claims
Any 3PL claiming to handle medical products should provide documented proof of FDA establishment registration, ISO 13485 certification, and GMP adherence. Verbal assurances are not sufficient in a regulated industry.
What to verify:
- FDA registration — Required for 3PLs acting as repackagers, relabelers, or initial importers. Verify status using the FDA's Establishment Registration & Device Listing database.
- ISO 13485 certification — The internationally recognized standard for medical device quality management systems. Verify certificates through the issuing accredited certification body or the IAF CertSearch database.
- GMP compliance — Required under 21 CFR Part 820 for entities performing repackaging, relabeling, or storage of medical devices.
If a provider cannot produce current certificates on request, or is evasive about which certifications apply to their warehouse versus their administrative operations, walk away.
Opaque or Shifting Pricing Structures
Hidden fees for receiving, storage minimums, special handling, returns processing, or labeling are common issues with 3PLs. A trustworthy provider should give a clear, itemized cost estimate based on your volume and service requirements before you sign.
Common hidden fees to watch for:
- Monthly storage minimums averaged $517 in 2025 — confirm this figure upfront before signing
- 48.6% of warehouses charge 1.5–3× the standard rate for inventory held beyond 30, 60, or 90 days
- Receiving fees vary widely depending on whether inventory is counted per pallet, per container, or per SKU
- Temperature-controlled storage, sterile packaging, and FDA-compliant labeling often carry separate special handling charges
Vague answers to direct pricing questions — or contracts that require extensive negotiation just to understand base rates — are signs of either disorganization or an intentionally confusing billing structure.
Refusal or Delay in Scheduling a Warehouse Tour
A 3PL managing medical products should welcome a facility walkthrough. You need to see actual storage conditions, cleanliness standards, temperature control if applicable, and how inventory is organized and tracked.
If scheduling a tour takes several weeks or requires unusual justification, the facility is likely not consistently maintained at the standard they advertise.
What to look for during a tour:
- Clean, organized storage areas with proper shelving and segregation
- Temperature and humidity monitoring systems (if applicable to your products)
- Clear labeling and inventory tracking processes
- Staff training documentation and compliance protocols visible on-site
No Real-Time Inventory Visibility or Outdated Technology
Without a modern Warehouse Management System (WMS) that integrates with your sales channels (Amazon, DTC website, etc.), you will have no reliable way to monitor stock levels, catch fulfillment errors, or track shipments in transit.
Required WMS capabilities:
- Real-time inventory tracking by SKU, lot, or serial number
- Barcode and UDI scanning for accurate tracking and serialization
- Integration with ecommerce platforms and sales channels
- Automated alerts for low stock levels or fulfillment errors
- Returns management workflows

Any provider that cannot demonstrate live dashboards or real-time order tracking during your evaluation is not ready to handle medical fulfillment at scale.
No Experience or References in the Medical or Healthcare Sector
Handling medical products requires hands-on experience with fragility, sterility, regulatory labeling, and international documentation. A 3PL without this background will have a steep learning curve — and you'll absorb the cost in errors and delays.
What to ask:
- Can you provide client references in the medical, healthcare, or life sciences space?
- What types of medical devices have you handled (Class I, Class II, durable medical equipment, disposable supplies)?
- Have you managed FDA recalls or regulatory audits for clients?
Follow up with those references to ask about compliance incidents, error rates, and responsiveness. Packaging and labeling mistakes are leading causes of medical device recalls, so ask specifically about their track record.
Weak Contract Terms with No Accountability Provisions
A contract that does not clearly define service level agreements (SLAs), liability for damaged or mis-shipped goods, insurance requirements, and exit clauses puts all risk on the shipper.
Review these elements before signing:
- Liability caps — IWLA Standard Contract Terms often limit liability to declared values per item or pound — confirm these limits don't fall far below your actual product value.
- Insurance coverage — Request annual proof of insurance and confirm coverage limits match your inventory's total value.
- Audit rights — Contracts should include provisions for facility and inventory audits.
- Exit clauses — Understand termination terms and how inventory will be handled if you switch providers.
Green Flags: What a Trustworthy Medical 3PL Looks Like
Red flags tell you when to walk away. These green flags tell you when you've found a partner worth trusting with your products.
Verified, Up-to-Date Compliance Documentation
A reliable medical 3PL will proactively share their FDA registration status, ISO 13485 certification, GMP compliance records, and HIPAA-safe handling protocols without being asked twice. This transparency signals genuine operational credibility.
What proactive documentation looks like:
- Current, dated certificates with visible expiration dates
- Certification body contact information for verification
- Clear documentation of which warehouse operations are covered under each certification
- Recent audit reports or FDA inspection results (if applicable)
Transparent Pricing and Scalable Service Structure
Look for providers who give clear per-unit or per-shipment estimates and explain their fee structure without ambiguity. Service tiers should scale with your business as volume grows or fluctuates seasonally — not lock you into rigid contracts that punish flexibility.
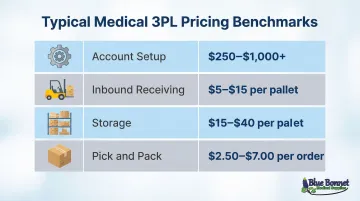
Typical pricing benchmarks:
| Fee Type | Typical Range (USD) | Notes |
|---|---|---|
| Account Setup | $250 – $1,000+ | One-time setup fees |
| Inbound Receiving | $5 – $15 per pallet | Includes put-away into storage |
| Storage | $15 – $40 per pallet | Monthly cost |
| Pick & Pack | $2.50 – $7.00 per order | Varies by number of items |
Medical-Specific Storage and Handling Protocols
Reliable providers maintain dedicated storage environments for sensitive medical items, with proper packing procedures that prevent damage to devices and supplies. They also have documented processes for handling returns — including product testing and restoration for items that can be requalified for resale.
Key capabilities:
- Temperature and humidity monitoring systems
- Segregated storage areas for different product categories
- Sterile packaging and FDA-compliant labeling processes
- Returns processing with inspection, testing, and restoration workflows
Direct Carrier Relationships and International Shipping Competency
Solid carrier relationships translate directly into cost savings. 3PLs often pass along 10-30% discounts off standard shipping rates through volume-based negotiations — savings a small or mid-size medical product company couldn't access on its own.
Beyond domestic shipping, look for demonstrated fluency with international customs documentation. Missing or incorrect paperwork is one of the most common reasons medical shipments stall at the border. A competent 3PL handles all three of these routinely:
- Certificates to Foreign Governments (CFG) — FDA-issued certificates confirming devices are legally marketed for U.S. export
- Commercial invoices — Prepared with complete merchandise descriptions, quantities, values, and correct Harmonized Tariff Schedule (HTS) codes
- Country of origin marking — Verified and compliant with U.S. Customs and Border Protection requirements
Questions to Ask Before You Sign
Before any contract conversation, run through these questions with every 3PL you're seriously considering. The answers — and how confidently they're delivered — will tell you a lot.
Operational Questions
- What is your order cut-off time for same-day shipping?
- What storage conditions do you maintain for sensitive medical items?
- How do you handle a fulfillment error or a damaged product claim?
- Will we have access to a live inventory dashboard?
- What ecommerce platforms does your WMS integrate with?
Compliance and Legal Questions
- Can you provide current copies of your FDA registration, ISO 13485, and GMP certificates?
- What does your liability coverage look like, and what are the per-incident limits?
- How do you handle a regulatory audit?
- Have you been subject to FDA inspections or warning letters?
Relationship and Communication Questions
- Will we have a dedicated account contact?
- How quickly do you respond to urgent issues?
- How do you communicate proactively when there's a problem with an order, before the customer calls us?
- Can you provide references from current medical product clients?
How Bluebonnet Medical Supplies Checks Every Box
Why Bluebonnet Medical Supplies Is Built for Medical 3PL
Bluebonnet Medical Supplies is a 3PL warehouse built specifically for medical product businesses. FDA clearance for medical packaging, ISO compliance, GMP compliance, and HIPAA-safe handling are foundational to how the facility operates — not optional upgrades layered on top of a general fulfillment workflow.
Bluebonnet treats each client as a business with its own workflows. That dedicated attention means issues get identified and resolved before they affect your customers or your compliance standing.
What's Included in the Service
- Storage and fulfillment with FDA-compliant handling
- Custom packaging and labeling, including sterile packaging options
- Domestic and international shipping with customs documentation support for medical exports
- Returns processing with product testing and restoration capabilities
- Direct carrier relationships that provide discounted shipping rates

If you're evaluating 3PL partners for your medical product business, reach out to info@bbmstx.com to discuss your specific logistics needs.
Frequently Asked Questions
What certifications should a 3PL have to handle medical products?
Medical product 3PLs should hold:
- FDA establishment registration (required if acting as repackager, relabeler, or initial importer)
- ISO 13485 certification for medical device quality management systems
- GMP compliance under 21 CFR Part 820
HIPAA-safe handling protocols are also essential for any business dealing with patient-related or health data-adjacent products.
How do I verify a 3PL's compliance credentials before signing?
Request current, dated certificate documents and cross-reference them with the issuing body. Confirm FDA registration through the FDA's Establishment Registration & Device Listing database and ISO 13485 status via IAF CertSearch. Never accept verbal claims or website badges as proof.
Is it normal to tour a 3PL warehouse before committing?
Yes, a facility visit is a standard and expected part of the evaluation process for any serious 3PL partnership. Any reluctance to schedule a walkthrough promptly is a warning sign, as it may indicate the facility is not consistently maintained at the standard they advertise.
What happens if a 3PL mishandles or loses my medical inventory?
Your contract should define liability limits and insurance coverage for damaged or lost goods. Review these clauses carefully before signing and ensure coverage is adequate relative to your product value. Standard IWLA contract terms often cap liability at specific amounts per item or pound, which may not cover high-value medical devices.
Can a general 3PL handle medical products if they're not certified?
A general 3PL can physically move items, but without FDA registration, ISO 13485, and GMP compliance, they cannot meet the handling and documentation standards regulated medical products require. The gap creates real legal exposure — recalls, customs holds, and regulatory enforcement are all possible outcomes.
How do I evaluate a 3PL for international medical product shipping?
Ask about their experience with medical device customs documentation, destination-country import requirements, and international carrier relationships. Confirm they can prepare Certificates to Foreign Governments (CFG) and are familiar with FDA export regulations under Section 801(e)(1).


