
Introduction
One bad fulfillment partnership can do more than hurt your margins — a failed audit, a compliance violation, or a mishandled return can cost you your FDA clearance. Medical devices and supplies aren't standard ecommerce inventory. They require sterile packaging, GMP storage protocols, and HIPAA-safe data handling that most logistics providers aren't equipped to deliver.
Getting this wrong is a regulatory problem, not just a shipping one.
Most "questions to ask a fulfillment company" guides are written for general ecommerce sellers. They focus on pricing, shipping speed, and technology integrations. But medical product businesses face a categorically different set of requirements that generic checklists don't address. The global medical device 3PL market reached $28.76 billion in 2024 and is projected to hit $47.01 billion by 2030 — yet many providers in this growing market lack the specialized credentials medical businesses actually need.
This guide provides a practical set of questions organized by category to help medical product businesses evaluate fulfillment partners before signing anything. These aren't generic logistics questions — they're compliance-first, risk-aware inquiries designed to protect your products, your regulatory standing, and your customers.
TLDR
- Verify certifications first (FDA, ISO, GMP, HIPAA) — non-compliant providers create direct regulatory liability for your business.
- Get itemized pricing upfront: receiving, storage, handling, and any minimum order penalty clauses.
- Probe operations directly: ask how they track inventory, resolve errors, and process returns.
- Test for scalability: carrier relationships, customs experience, and how they handle demand spikes.
Why Medical Product Fulfillment Demands a Different Checklist
Medical products — from diagnostic devices to surgical supplies — are regulated goods. Under FDA regulations, manufacturers remain legally responsible for the compliance of their outsourced fulfillment partners. The FDA's Quality System Regulation explicitly governs "the design, manufacture, packaging, labeling, storage, installation, and servicing of all finished devices." If your 3PL fails to maintain compliance, you face the regulatory consequences.
Consider the real-world impact: Exactech recalled 80% of its knee and ankle replacements due to defective packaging bags that allowed oxidation — a failure that ultimately pushed the company into bankruptcy. This wasn't a manufacturing defect; it was a packaging and storage failure. Between 2019 and 2024, seal-integrity and packaging issues accounted for 2-4% of all FDA medical product recalls annually.
Medical fulfillment compliance operates across multiple layers:
- FDA clearance for packaging and labeling operations
- GMP protocols governing storage conditions and handling procedures
- ISO 13485 quality standards for medical device quality management systems
- HIPAA-safe data handling when patient information flows through the supply chain

When vetting a fulfillment partner for medical products, compliance credentials come first. Any provider worth considering should be able to produce documentation on demand — certifications, audit records, and standard operating procedures specific to regulated goods.
Questions to Ask About Compliance, Certifications, and Product Handling
Compliance credentials are non-negotiable for medical fulfillment. Any provider that can't document them clearly should be off your list before you dig into pricing or logistics.
Are you FDA cleared, ISO certified, and GMP compliant?
FDA clearance for medical packaging services, ISO compliance, and GMP compliance are not optional — they are baseline requirements. Don't accept verbal confirmation; ask candidates to provide documentation.
Each certification covers distinct compliance areas:
- ISO 13485 speaks to quality management systems and is the international standard for medical device QMS. The FDA's new Quality Management System Regulation (QMSR), effective February 2, 2026, incorporates ISO 13485 by reference.
- GMP (Good Manufacturing Practice) governs storage and handling practices under 21 CFR Part 820, including requirements for stock rotation when product quality deteriorates over time.
- FDA clearance covers packaging operations. Under 21 CFR Part 807, establishments involved in contract packaging, repackaging, or relabeling must register annually with the FDA.

A qualified provider should be able to explain how each standard is implemented in their facility and provide copies of current certifications.
How do you store and handle sensitive or fragile medical products?
Medical storage requirements vary significantly by product type. Generic pallet racking designed for consumer goods won't cut it. Ask specific questions:
- Do you have temperature-controlled zones for products requiring specific storage conditions?
- What specialized shelving or racking systems do you use for fragile or sterile items?
- Are warehouse staff trained specifically for medical-grade handling protocols?
- How do you prevent cross-contamination between product types?
FDA storage regulations (21 CFR 820.150) require control of storage areas to prevent mix-ups, damage, or deterioration. A strong answer will include specific protocols — trained staff, segregated storage zones, and documented procedures. Vague responses about "careful handling" are a red flag.
Can you demonstrate HIPAA-safe data handling practices?
If your business involves any patient-linked data — even in labeling or order records — HIPAA compliance in data handling is required. When a 3PL processes identifiable patient health information (PHI), they act as a "Business Associate" under HIPAA.
Ask these specific questions:
- Will you sign a Business Associate Agreement (BAA)?
- How is customer and order data stored, and who has access to it?
- Have you had any data breaches or security audits?
- Do you encrypt PHI both in transit and at rest?
The HHS Office for Civil Rights clarifies that the "conduit exception" applies only to transmission-only services like UPS. A 3PL that stores or processes ePHI on a persistent basis does not qualify for this exception and must sign a BAA.
Do you have experience fulfilling products for companies like mine?
Ask whether they currently work with medical device manufacturers, online medical supply retailers, or Amazon sellers in the medical category. A provider with relevant experience understands the compliance landscape, not just general fulfillment operations.
When evaluating experience, request:
- References from clients in similar product categories
- Case studies showing how they've handled medical-grade documentation or traceability
- Examples of compliance issues they've navigated on a client's behalf
A 3PL that primarily serves apparel or consumer electronics may lack the documentation practices and handling protocols your products require.
Questions to Ask About Pricing, Contracts, and Hidden Fees
Competitive rates on a 3PL's website don't always reflect what you'll actually pay. Many providers advertise low base pricing but embed significant costs elsewhere in the contract — and up to 20% of freight invoices contain billing inaccuracies. Before you sign anything, get every fee in writing.
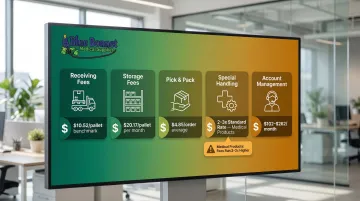
What is the full fee breakdown — including receiving, storage, pick-and-pack, and special handling?
Request a written itemized quote that covers every charge category:
| Fee Component | What to Ask |
|---|---|
| Receiving fees | How is this calculated — per pallet, per carton, or per SKU? The 2025 benchmark is $10.52 per pallet. |
| Storage fees | What is the monthly rate per pallet or per cubic foot? Rates have doubled since 2016; the current average is $20.17 per pallet/month. |
| Pick and pack | What is the base fee per order? B2B orders average $4.85 per order due to higher complexity. |
| Special handling | What additional fees apply for medical products requiring sterile handling, temperature control, or lot tracking? |
| Account management | Are there monthly platform fees or account maintenance charges? The average ranges from $102.88 to $261.80/month. |
For medical products, special handling fees — sterile packaging, cold storage, lot tracking — often run two to three times the standard rate. Get exact figures for each service your products require, not a generic estimate.
Are there minimum monthly order volumes, and what happens if I fall short?
Many 3PLs require minimum order quantities (MOQs). The average monthly minimum spend requirement increased to $517 in 2025, up from $337.50 in 2024. For medical product businesses with variable demand — especially those launching new SKUs or expanding into international markets — penalties for falling below minimums can erode margins.
Get the exact minimum threshold and penalty structure in writing. Some providers charge the difference between your actual volume and the minimum; others apply a flat percentage penalty. Either way, model out your worst-case month before agreeing to the terms.
What does the contract term look like, and is there flexibility to exit?
Long-term contracts carry risk if the partnership underperforms. Before committing, clarify:
- Contract length (month-to-month, annual, multi-year)
- Notice periods for termination (30 days, 60 days, 90 days)
- Penalties for early exit
- Whether there is a trial or evaluation period before a long-term commitment is required
Medical product businesses should negotiate flexibility, especially when first outsourcing fulfillment or testing a new provider.
What is your policy on inventory shrinkage, loss, and product damage?
Shrinkage — goods lost, stolen, or damaged in the warehouse — is an inherent risk in any 3PL environment. Before signing, get clear answers on:
- The provider's shrinkage allowance (the acceptable percentage of lost goods before they owe reimbursement)
- What their liability policy covers and any coverage caps
- How they document and report shrinkage incidents
For medical products, even small losses can have compliance implications, especially for serialized or lot-tracked items. If your inventory includes high-value or serialized items, confirm that the liability cap reflects replacement cost — not just the wholesale price the provider assigns.
Questions to Ask About Operations, Technology, and Returns
Once a provider passes the compliance and pricing checks, these questions determine whether their day-to-day systems are built to support your business reliably.
How do you track inventory, and will I have real-time visibility?
Real-time inventory visibility is essential for medical product businesses managing multiple SKUs, regulatory lot numbers, or expiration dates. Ask:
- Does your warehouse management system (WMS) support SKU-level tracking?
- Can I access inventory data through a dashboard or direct integration with my selling channels (Amazon, Shopify, etc.)?
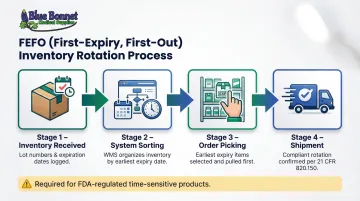
- Does the system support FEFO (First Expiry, First Out) inventory rotation?
FEFO (First-Expiry, First-Out) ensures stock with the earliest expiration date ships first. 21 CFR 820.150 requires time-sensitive products be stored to enable proper rotation — FEFO is the accepted operational standard for meeting that requirement.
What is your order accuracy rate, and how do you handle errors?
Order accuracy directly affects customer relationships and, in the medical space, can have patient safety implications. According to the WERC 2022 DC Measures report, the industry median for order-picking accuracy is 99.20%, while best-in-class operations achieve ≥99.85%.
Ask for:
- The provider's documented pick error rate
- Whether they have an error penalty policy — meaning they absorb correction costs when mistakes are their fault
- How errors are reported and tracked
If a provider won't share accuracy data or own their mistakes, that tells you everything you need to know before the returns conversation.
How do you handle returned medical products — and can items be tested or restored?
Medical product returns require more than a standard "restock and resell" workflow. Ask whether the provider can:
- Assess returned items for condition and functionality
- Perform basic product testing to verify usability
- Restore resalable items to inventory with proper documentation
Not all 3PLs offer this capability. For medical businesses, returns processing with testing and restoration can prevent significant losses on returned inventory while maintaining compliance with quality standards.
Questions to Ask About Shipping, Scalability, and Customer Support
After compliance, pricing, and operations, these final questions assess whether the provider can grow with your business and communicate clearly when problems arise.
What carrier relationships do you have, and can you access discounted rates?
The best fulfillment partners have direct carrier relationships that enable negotiated shipping rates — savings they can pass to clients. Ask:
- Which carriers do you work with (UPS, FedEx, USPS, regional carriers)?
- Can you support both domestic and international shipping?
- Do your medical product customers receive carrier-specific discounts?
Carrier discounts can represent 10-30% savings on shipping costs, which impacts your margins and competitiveness.
Can you handle international medical shipments and customs compliance?
International medical device and supply shipments involve customs documentation, import regulations, and product-specific compliance in destination countries. In a single 2021 operation, U.S. Customs seized 26,477 pairs of misbranded contact lenses valued at $479,082 because they lacked proper FDA clearance and labeling.
Ask whether the provider has:
- Experience with international medical fulfillment
- Knowledge of destination country requirements (EU MDR, Health Canada MDEL, TGA ARTG registration)
- Processes to ensure products don't get delayed or seized at customs
- Accurate Harmonized System (HS) code assignment for medical devices
For EU shipments, importers must verify CE marking and ensure the importer draws up a declaration of conformity. For Canada, Class II-IV devices must have a Medical Device Licence. Your 3PL must understand these requirements.
How will you communicate with me day-to-day, and who is my point of contact?
When a shipment gets flagged or a compliance question surfaces, you can't afford to wait two days for a ticket response. Ask:
- Will I have a named account manager?
- How quickly are issues escalated?
- Is support available during critical periods (product launches, holiday peaks)?
For medical product businesses, a faceless ticketing system falls short when compliance or shipment issues arise. You need direct access to someone who knows your products and can resolve problems fast.
How Bluebonnet Medical Supplies Can Help
Bluebonnet Medical Supplies is a 3PL built specifically for businesses that need compliance assurance, not just logistics throughput. Every client gets a solution tailored to their product type, workflow, and regulatory requirements — not a generic system retrofitted to fit.
Bluebonnet's credentials align directly with the questions outlined in this guide:
- FDA-cleared medical packaging services
- ISO compliance for quality management systems
- GMP compliance for storage and handling protocols
- HIPAA-safe handling for patient-linked data
The Cedar Park, Texas facility maintains dedicated storage controls for sensitive medical items, including temperature-managed areas and handling protocols designed for regulated products. Key service capabilities include:
- Storage and handling designed specifically for medical products
- Packing and FDA-compliant shipping for domestic and international orders
- Returns processing, product testing, and restoration services
- Direct carrier relationships offering discounted shipping rates
- Custom solutions for Amazon sellers, direct-to-consumer medical brands, and international distributors

Reach out to Bluebonnet at info@bbmstx.com to talk through your fulfillment needs before you commit to a contract.
Conclusion
Signing a fulfillment contract is one of the most consequential operational decisions a medical product business makes. The questions in this guide aren't about being difficult — they're about protecting your products, your compliance standing, and your customers.
Compliance credentials are the filter, not the finish line. A provider who passes the compliance check still needs to demonstrate pricing transparency, operational reliability, and the ability to communicate when things go wrong.
The right fulfillment partner should evolve with your business. As your product catalog grows, your sales channels expand, or your international footprint develops, revisit whether your partner is still meeting the bar. Build that review into your annual operations calendar — your fulfillment setup on day one rarely looks the same as what you need in year three.
Frequently Asked Questions
What do fulfillment companies do?
Fulfillment companies receive, store, pick, pack, and ship products on behalf of businesses. Medical fulfillment companies add compliance-specific capabilities like FDA-cleared packaging, GMP storage protocols, and returns processing for regulated products.
What are the 7 steps of the order fulfillment process?
The standard stages are receiving inventory, storage, order processing, picking, packing, shipping, and returns handling. For medical products, each stage includes additional compliance checkpoints such as lot tracking, expiration date verification, and documentation.
What are some good supply chain questions to ask a fulfillment partner?
Ask about inventory visibility, carrier relationships, order accuracy rates, error resolution processes, and scalability policies. For medical businesses, also ask about compliance certifications (FDA, ISO, GMP), HIPAA handling practices, and international customs experience.
What certifications should a medical product fulfillment company have?
Look for FDA clearance for medical packaging, ISO 13485, GMP compliance, and HIPAA-safe data handling practices. These certifications confirm the provider is equipped to manage regulated products correctly.
What should I look for in a fulfillment contract before signing?
Review the full fee structure, minimum order requirements, contract length and exit terms, shrinkage liability policies, and whether compliance obligations—including special handling fees for medical products—are explicitly documented.
Can a regular 3PL handle medical products?
Most general 3PLs are not equipped to handle medical products compliantly. They typically lack FDA clearance, GMP protocols, and specialized storage required for regulated medical devices. Partnering with an unqualified provider puts you at risk of regulatory violations, product delays, and potential fines.


