
Introduction
Medical supplies kitting is the process of assembling multiple medical products or components into a single, ready-to-ship unit under one SKU, handled in a compliant warehouse environment.
For medical product businesses, device manufacturers, Amazon sellers, and online medical supply retailers, kitting carries compliance and safety obligations governed by FDA, ISO, GMP, and HIPAA standards — not just logistical ones.
Unlike standard ecommerce kitting, medical supplies kitting operates under strict regulatory oversight. A 2024 FDA guidance clarifies that convenience kits are themselves medical devices. The businesses that assemble them are legally classified as manufacturers, not just distributors. This means every step — from component inspection to labeling — must meet manufacturing-grade quality standards.
Understanding those standards is what separates kitting done right from kitting that creates liability. This article breaks down what medical supplies kitting is, how the process works step by step, what makes it different from general kitting, and what compliance requirements apply at each stage.
TLDR
- Medical supplies kitting pre-assembles multiple products into compliant, labeled, ready-to-ship kits—cutting fulfillment time and reducing picking errors
- Medical kitting requires FDA, ISO, GMP, and HIPAA compliance that standard fulfillment workflows aren't built to handle
- Every kit moves through a defined sequence: design, inspection, assembly, labeling, SKU creation, and compliant storage
- Lot tracking, expiration management, and sterile handling determine whether a kit is legally shippable—not just functional
- Outsourcing to a certified 3PL allows businesses to fulfill orders faster while staying compliant
What Is Medical Supplies Kitting?
Medical supplies kitting is the practice of grouping related medical products into a single pre-packaged unit sold or fulfilled as one SKU. Think wound care supplies, diagnostic components, procedure packs, or device accessories—bundled together under one label.
Kitted products arrive complete, correctly assembled, properly labeled, and ready for clinical, retail, or patient use. This removes the need for individual item picking or last-minute assembly at the point of fulfillment, cutting errors and speeding up delivery.
How Medical Kitting Differs from General Ecommerce Kitting
While both involve assembling multi-item units, medical kitting requires:
- Compliance with regulated packaging standards
- Proper storage conditions (temperature, humidity)
- Lot number tracking and expiration date management
- Sterility protocols where applicable
- FDA-cleared packaging credentials
None of these requirements apply to standard consumer kitting. Under 21 CFR 807.20, 3PLs that assemble, repackage, or relabel medical kits are legally classified as manufacturers, triggering full Quality Management System requirements. That regulatory weight is also what separates medical kitting from a packaging decision.
Kitting vs. Packaging: Understanding the Distinction
Packaging refers to the container or wrapper around a single product. Kitting is the act of combining multiple distinct products into a single sellable or shippable unit. Packaging is one component of the kitting process, not the same thing.
A sterile wound care kit, for instance, might contain gauze pads, adhesive tape, antiseptic wipes, and gloves—each individually packaged, then assembled into one unit with a primary label. The packaging protects each item; the kitting creates the product.
Why Kitting Is Critical in the Medical Supply Industry
The Compliance Risk
Medical supply businesses operating in the US must adhere to FDA regulations for device packaging and labeling, GMP standards for handling, and HIPAA-safe protocols for patient-related products. Kitting that skips these requirements creates real liability — failed customs clearance, compliance violations, and product recalls.
FDA Import Alert 89-04 authorizes Detention Without Physical Examination (DWPE) for devices from manufacturers whose facilities don't conform to Current Good Manufacturing Practices. A single unapproved component or packaging failure can get the entire kit detained at the border.
The Operational Driver
Medical product businesses—especially those selling on Amazon, through their own websites, or exporting internationally—need to fulfill orders quickly and accurately. Pre-assembled kits reduce pick-and-pack time, minimize the risk of missing components, and enable faster shipping.
Research shows that a standard 97% warehouse picking accuracy rate (a 3% error rate) bleeds between $705,000 and $1.4 million annually in hard costs per 500,000 orders. Kitting consolidates multiple SKUs into one unit, sharply cutting picking errors.
The Patient Safety Factor
Healthcare providers and patients rely on kits arriving complete and undamaged. An incorrectly kitted procedure pack or missing component in a diagnostic kit can create clinical risk, not just a return. Operating room delays cost hospitals approximately $153 per minute, making accuracy essential.
What Goes Wrong Without Proper Medical Kitting
- Shipments held at customs due to improper labeling
- Returned orders due to missing components
- Compliance violations from unregulated packaging
- Higher per-unit fulfillment costs from inefficient individual item picking
- Product recalls triggered by packaging failures
How the Medical Supplies Kitting Process Works
Medical kitting follows a defined sequence: kit design and compliance planning, component receiving and inspection, assembly, labeling, SKU assignment, and storage. Each stage must be documented and controlled to meet regulatory requirements.
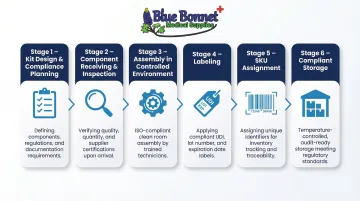
Step 1: Define the Kit and Confirm Compliance Requirements
The process begins by determining:
- Which products go into the kit
- The intended end use (retail, clinical, or export)
- Applicable compliance requirements for that product category
This includes FDA labeling rules, lot tracking obligations, sterility requirements, and any specific packaging standards for the product category. Under 21 CFR 801.30(a)(11), individual devices packaged within a convenience kit are exempt from bearing their own UDI, provided the kit's primary label bears a compliant UDI—which reduces labeling costs.
Step 2: Receive and Inspect Components
Incoming components are received at the warehouse and inspected for:
- Damage and completeness
- Compliance with specifications
- Lot numbers and expiration dates (logged for traceability)
- Quantities verified against the kit bill of materials
Under 21 CFR 820.45, manufacturers must ensure labeling and packaging have been examined for accuracy prior to release. Non-conforming items are flagged before assembly begins.
Step 3: Assemble the Kit in a Controlled Environment
Kitting takes place in a warehouse environment designed for sensitive medical items:
- Appropriate storage conditions (temperature, humidity)
- Clean handling protocols
- Organized kitting stations
For regulated products, assembly follows documented work instructions to ensure repeatability. ISO 13485 Clause 7.5.11 requires organizations to document procedures for preserving product conformity during processing, storage, handling, and distribution.
Step 4: Label, Assign a SKU, and Prepare for Fulfillment
Each completed kit receives compliant labeling:
- Product name
- Lot number
- Expiration date
- Country of origin
- Regulatory markings as required
The kit is assigned a unique SKU in the inventory system and stored in a designated location ready for order fulfillment. For medical product businesses that don't want to manage this infrastructure in-house, partnering with a qualified 3PL—one holding FDA cleared medical packaging credentials and operating under ISO and GMP compliance, like Bluebonnet Medical Supplies—keeps this stage fully compliant without adding internal overhead.
Key Factors That Affect Medical Kitting Quality and Compliance
Not all kitting operations are equal — and in medical supply chains, the gaps show up as compliance failures, recalled products, or customs holds. These are the factors that separate reliable kitting from risky kitting:
Lot tracking and expiration management: Kits must follow FEFO (First Expired, First Out) picking logic, as mandated by industry standards. Mixed lot numbers or near-expiry components create traceability gaps and direct regulatory exposure.
Storage and handling conditions: Temperature-sensitive, sterile, or fragile medical products require warehouse environments built to handle them. Standard consumer warehouses are not equipped for this and can compromise product integrity before the kit is even shipped.
Regulatory labeling accuracy: FDA-regulated products require specific label elements. Incorrect, missing, or non-compliant labeling on a kit is a customs violation and a recall risk for exported or commercially sold medical supplies.
Volume and throughput: Kitting efficiency depends on adequate staffing, defined work instructions, and inventory systems that automatically update component-level counts during assembly. Manual tracking at high volumes creates errors.
Returns and restocking protocols: Returned kits require a defined inspection process — component separation, assessing what can be restocked, and disposing of non-conforming items properly. Not all returned medical products are eligible for re-kitting.

Common Misconceptions About Medical Supplies Kitting
Misconception: Medical Kitting Is the Same as Regular Warehouse Kitting
The compliance layer—FDA cleared packaging, lot tracking, temperature-controlled storage, HIPAA-safe handling—makes medical kitting a regulated process, not just a packing activity. Under 21 CFR 820.3, 3PLs that assemble kits are legally manufacturers subject to Quality Management System regulations. Treating it as generic fulfillment creates legal and product quality risk.
Misconception: Kitting Is Only Relevant for Large-Volume Operations
Even small medical product businesses benefit from kitting. The reduction in per-order fulfillment time, picking errors, and customs complications applies at any scale. The threshold is whether the product is being sent to customers in a complete, compliant, ready-to-use form.
Misconception: Any 3PL Can Handle Medical Kitting
Most general 3PLs are not equipped for medical product handling. Before selecting a kitting partner, medical product businesses should confirm the provider has:
- FDA cleared packaging capabilities
- GMP-compliant assembly processes
- HIPAA-safe handling protocols
- Lot tracking and chain-of-custody documentation
The consequences of skipping this vetting are well-documented. DeRoyal's mislabeled anesthetic packs contained 1% lidocaine mislabeled as 0.5% bupivacaine, risking fatal overdose. Alcon's incomplete sterile seals compromised the sterile barrier in ophthalmic procedure packs.

Both incidents resulted in Class I recalls and immediate market withdrawals — outcomes that proper kitting compliance is specifically designed to prevent.
Conclusion
Medical supplies kitting is a structured, compliance-driven process that assembles multiple products into ready-to-ship units. Done well, it reduces fulfillment errors, speeds up shipping, meets regulatory requirements, and ensures products arrive complete and fit for their intended medical use.
Medical kitting is not the same as consumer ecommerce fulfillment. Businesses that treat it that way risk compliance failures, customs delays, and product integrity issues. The right warehouse partner brings certifications, purpose-built processes, and regulatory knowledge that general fulfillment providers simply don't have.
What to look for in a kitting partner:
- FDA clearance for medical packaging
- ISO and GMP compliance for quality control
- Experience with customs documentation for international shipments
- Lot tracking and serialization capabilities
- Returns processing with product restoration
For medical product businesses looking to outsource kitting while maintaining compliance, Bluebonnet Medical Supplies offers FDA-cleared, ISO and GMP compliant kitting and fulfillment services built specifically for medical products. Products get packed right, shipped right, and handled with care — so you can focus on growing your business, not managing logistics. Reach out at info@bbmstx.com to discuss your kitting needs.
Frequently Asked Questions
What is the kitting process?
Kitting involves gathering individual product components, assembling them into a single pre-packaged unit, assigning a new SKU, and preparing it for storage and fulfillment. In medical supplies, this process also includes inspection, lot tracking, and compliant labeling.
What is the difference between kitting and fulfillment?
Kitting is the pre-assembly step that creates the ready-to-ship unit before or during fulfillment. Fulfillment is the broader process of receiving an order, picking, packing, and shipping it — kitting speeds that up by reducing the number of picks required per order.
What is the difference between kitting and packaging?
Packaging refers to the container or material enclosing a single product. Kitting is the act of combining multiple distinct products into one sellable or shippable unit. Packaging is one element used within the kitting process, not a synonym for it.
What is the purpose of kitting?
Kitting reduces fulfillment time and errors by pre-assembling multi-product orders, and improves the customer experience by delivering complete, ready-to-use sets. In the medical industry specifically, it ensures all required components arrive together in a compliant, properly labeled package.
What does a kitting specialist do?
A kitting specialist assembles kits per defined work instructions, inspects components for quality and completeness, applies labels and documentation, and tracks lot numbers and expiration dates. Each finished kit is verified against required standards before it enters inventory.
What is fulfillment kitting?
Fulfillment kitting (also called full kitting) refers to kitting performed as part of the warehouse fulfillment process. Components are assembled into a single SKU in the warehouse so that when an order is placed, only one item needs to be picked and shipped rather than multiple individual components.


