
Introduction
Medical supply businesses face a critical decision when structuring their product offerings: should you pre-assemble items into kits, or group them together as bundles? Both strategies can streamline operations and drive sales, but they work differently. Picking the wrong one can create compliance headaches, inefficient warehousing, or missed revenue.
This decision carries particular weight for medical product businesses because it directly affects SKU management, FDA-compliant packaging requirements, fulfillment speed, and customer experience across channels like Amazon and DTC websites. Get it right, and your entire logistics chain runs more smoothly.
TLDR
- Kitting assigns one SKU to a pre-assembled unit, cutting fulfillment time by up to 80%
- Bundling groups separate products for promotions and can boost average order value by 20-30%
- Regulated medical products require FDA/GMP-compliant assembly for kitting; bundling carries lower compliance overhead
- Over 90% of medical procedures use device kits — 250+ million are used in the U.S. annually
- Most businesses benefit from both: kitting for core products, bundling for promotions
Kitting vs. Bundling: Quick Comparison
| Factor | Kitting | Bundling |
|---|---|---|
| Purpose | Pre-assemble items into single units for fulfillment efficiency | Group products for promotional offers to increase sales |
| SKU Management | Creates one new SKU; simplifies inventory tracking | Each item keeps individual SKU; bundle is marketing construct |
| Fulfillment Process | Pre-assembled before orders arrive; faster pick-and-pack | Items picked separately at order time; slower during peaks |
| Compliance (Medical) | Requires FDA/GMP assembly, proper labeling, sterile handling | Lower complexity; products not physically merged |
| Best For | High-volume recurring sets, subscription boxes, procedure kits, FBA prep | Seasonal promotions, clearing slow inventory, testing combinations |

What is Kitting?
Kitting combines multiple individual components into a single pre-packaged unit — one SKU, shipped as one item. Unlike bundling, kitting happens before an order is placed, typically during scheduled assembly runs at the warehouse.
Because kits are ready before orders arrive, fulfillment teams pick one item instead of multiple, cutting packing time and reducing picking errors. Implementing advanced kitting systems has reduced kitting times by up to 80% for medical device companies, while Merit Medical Systems decreased assembly time from hours to minutes and cut picking labor by 50% using automated kitting solutions.
Kitting Requirements for Medical Products
Medical kitting carries requirements that set it apart from general retail kitting. Over 90% of medical procedures use kits, with more than 250 million medical and surgical kits used annually in the United States.
Common examples include surgical procedure kits, wound care sets, diagnostic test kits, and first aid kits — all requiring assembly in compliant environments.
Medical device kitting must adhere to strict regulatory standards:
- FDA packaging guidelines under 21 CFR 820 (Quality Management System Regulation)
- GMP (Good Manufacturing Practice) standards for assembly processes
- Sterile or controlled-environment assembly depending on product classification
These compliance standards also dictate the physical assembly environment. Low-risk, non-sterile items can be assembled in general settings, while non-sterile medical components require white rooms with filtered air and controlled temperature/humidity. Sterile or implantable devices need ISO 14644-1 certified clean rooms.

Kitting Variations
- Manufacturer-level kitting — products leave the factory as complete kits before entering distribution
- Outsourced kitting — a compliant 3PL partner handles assembly, maintaining regulatory compliance without requiring in-house clean room facilities
- On-demand kitting — units are assembled as orders arrive, offering flexibility for customized configurations
For medical product businesses without clean room infrastructure, outsourced kitting through an FDA/GMP-compliant 3PL like Bluebonnet Medical Supplies covers both operational efficiency and regulatory compliance.
Use Cases of Kitting
Kitting fits best in specific scenarios where efficiency and consistency matter most:
- Recurring subscription shipments like monthly diabetic care supplies or wound care refills
- Amazon FBA prep where pre-kitted units simplify inbound requirements and reduce per-unit fees
- New product launch kits showcasing complete product systems
- Procedure-specific surgical kits for healthcare facilities
- High-volume repeat orders where the same combination ships frequently
The efficiency gains are measurable. Reorganizing inventory based on kitting frequency led to 36-49% reductions in kitting times for high-demand parts, while Performance Health increased picker productivity by 97% and reduced required labor by 50% using box-first kitting automation.
What is Bundling?
Bundling is a sales and marketing strategy where two or more separate products are offered together at a combined or discounted price—without physically merging them into a new SKU. Each item remains individually stored, tracked, and picked at order time.
The primary purpose is marketing-driven: bundling raises average order value, moves complementary or slow-moving products, and creates a stronger perceived value for customers. For example, a medical supply company might bundle a blood pressure monitor with replacement cuffs and batteries at a promotional price, creating a complete solution that encourages larger purchases.
Inventory and Fulfillment Implications
Since products retain individual SKUs and aren't pre-assembled, bundling is easier to set up and modify than kitting. You can create or discontinue bundles without warehouse restructuring. However, fulfillment teams must pick multiple items per order, which can slow operations during peak periods when order volume spikes.
That operational flexibility comes at a cost to fulfillment speed — a trade-off worth weighing carefully before scaling bundle volume.
Use Cases of Bundling
Bundling fits best when sales flexibility and inventory management are priorities:
- Limited-time promotions like holiday gift bundles or seasonal health kits
- Pairing best-sellers with slow movers to improve inventory turnover—bundled products turn over 30% faster than individual items
- Testing new product combinations before investing in kitting infrastructure
- Clearing excess inventory by creating attractive packages that move slow-moving stock
- Condition-specific bundles like "home health starter kits" grouping thermometers, pulse oximeters, and blood pressure monitors
The numbers back this up. Strategic bundling increases average order value by 20-30%, and McKinsey research puts sales gains at 20% with profit increases of 30%. A 2025 DME retailer case study recorded a 20% AOV increase and 15% margin improvement within six months of launching condition-specific bundles.

In practice, this looks like an online medical supply retailer offering a "home health monitoring bundle" — a thermometer, pulse oximeter, and blood pressure monitor at a discounted rate. Each item ships from existing inventory with no warehouse restructuring. The bundle appears as a single product listing, but the warehouse management system picks three individual SKUs when the order comes in.
Kitting vs. Bundling: Which Strategy Fits Your Product Line?
Choosing between kitting and bundling requires evaluating several factors specific to your business model and product characteristics.
Choose Kitting If Your Priority Is Operational Efficiency
For medical product businesses with high-volume, recurring orders for multi-component products, kitting dramatically reduces fulfillment time, minimizes errors, and lowers per-order shipping costs.
Kitting wins when:
- You ship the same product combinations repeatedly
- Fulfillment speed is critical to customer satisfaction
- You need to optimize dimensional weight for shipping (more on this below)
- You sell through Amazon FBA where pre-assembled units simplify inbound requirements
- Your products require sterile or controlled assembly anyway
Kitting consolidates multiple items into one optimized package, reducing dimensional weight charges. With carriers using a DIM divisor of 139 for domestic shipments and rounding every fractional inch up (effective August 2025), even small packaging improvements deliver significant savings.
A kit containing five items shipped in one 12×10×8 box (960 cubic inches, DIM weight 7 lbs) costs far less than five separate 6×4×3 boxes — each only 72 cubic inches, but billed as five individual shipments.
Choose Bundling If Your Priority Is Sales Flexibility
Bundling is the lower-effort entry point for businesses testing product combinations, running promotions, or clearing excess inventory without disrupting warehouse operations or incurring kitting assembly costs.
Bundling wins when:
- You want to test product combinations before committing to kitting
- You run frequent promotional campaigns with changing offers
- You need to move slow-moving inventory quickly
- Setup speed matters more than long-term efficiency
- Your products don't require special assembly environments
Bundling slow-moving items with best-sellers creates appealing packages that help liquidate excess inventory and maximize residual value. This "anchor bundle" approach converts stagnant inventory into cash while using popular items to draw customers.
The Compliance Factor for Medical Product Businesses
Kitting regulated medical devices or supplies requires FDA-cleared packaging environments, proper documentation, and sterile handling protocols. Under 21 CFR 820.3, a "manufacturer" includes any person who assembles or processes a finished device — covering contract sterilization, relabeling, and repacking.
CGMP compliance is required for both constituent parts and the overall kit if any of the following apply:
- Products are repackaged or relabeled from independently marketed items
- Kit labeling modifies the intended use of constituent parts
- Assembly involves sterile or controlled handling environments
Bundling carries less compliance complexity since products keep their original form and packaging. Each item retains its original labeling and regulatory status, simplifying documentation requirements.
Critical consideration: Businesses must verify their fulfillment partner can support FDA/GMP-compliant kitting before implementing this strategy. Partners like Bluebonnet Medical Supplies maintain FDA-cleared medical packaging capabilities and ISO compliance, ensuring kitted products meet regulatory requirements without requiring in-house clean rooms.
Can You Use Both?
Many medical product businesses use kitting for core product lines and bundling for promotional campaigns — a hybrid approach that delivers both operational efficiency and marketing flexibility.
How it works:
- Kitted products become permanent catalog SKUs with consistent, optimized fulfillment
- Bundles layer existing products — including kits — into time-limited promotional offers
- Warehouse operations remain streamlined because core kitting processes don't change
- Marketing teams gain flexibility to test offers without disrupting fulfillment
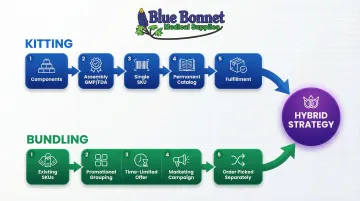
For example, you might maintain a kitted "diabetic care essentials" product (glucose meter, test strips, lancets, control solution) as a permanent SKU, then create seasonal bundles pairing this kit with a carrying case and logbook during holiday promotions.
Conclusion
There's no universal winner between kitting and bundling—the right choice depends on your goals, product type, and sales channels.
- Kitting wins on operational efficiency, compliance-ready assembly, and long-term cost reduction
- Bundling wins on marketing flexibility, low setup cost, and inventory agility
For medical product businesses selling regulated supplies through Amazon, DTC, or internationally, the compliance dimension adds real weight to this decision. Kitting requires FDA/GMP-compliant assembly environments and proper documentation, while bundling maintains simpler regulatory requirements since products aren't physically reconfigured.
The most successful medical supply businesses often use both strategies: kitting for high-volume core products where efficiency matters most, and bundling for promotional campaigns where flexibility drives sales. Partnering with a compliant 3PL that handles kitting, packaging, fulfillment, and shipping—like Bluebonnet Medical Supplies—means you can pursue either strategy (or both) without compromising FDA, GMP, or HIPAA-safe handling standards.
Want to figure out which approach works for your product line? Reach out to Bluebonnet Medical Supplies at info@bbmstx.com to talk through kitting and bundling options that fit how your business actually operates.
Frequently Asked Questions
What is the difference between kitting and bundling?
Kitting physically combines products into a single pre-assembled SKU for fulfillment efficiency, while bundling groups products together under a promotional offer where each item retains its own SKU and is picked separately at order time.
What is an example of product bundling?
A common example is a "home health monitoring bundle" pairing a blood pressure monitor, pulse oximeter, and thermometer at a discounted combined price. Each item ships from its own inventory slot — no pre-assembly required.
Can kitting and bundling be used together?
Yes, many businesses run both at once: kitting for core high-volume product lines where fulfillment efficiency matters most, and bundling for seasonal promotions or campaigns that don't need warehouse restructuring.
Does kitting create a new SKU for the assembled product?
Yes, once components are assembled into a kit, the kit receives its own unique SKU tracked as a single inventory unit. This makes reorder points, stock counts, and fulfillment workflows easier to manage.
Does kitting reduce shipping costs for medical product businesses?
Yes. Consolidating multiple items into one package reduces dimensional weight charges — a real advantage for businesses shipping via Amazon FBA or sending medical supplies internationally, where carriers price by package size and weight.
Do medical product kits require special compliance for assembly and packaging?
Yes. Kitting regulated medical devices or supplies must meet FDA packaging guidelines and GMP standards, with assembly done in a controlled, compliant environment. Choosing a certified 3PL partner is critical to staying compliant.


