
Introduction
In operating rooms and emergency departments, every second counts. Surgeons and nurses work under intense pressure — scrambling to locate a missing instrument mid-procedure isn't just frustrating, it's a patient safety risk. Clinical staff spend an average of 32% of their shift searching for supplies, time that should go toward direct patient care.
Medical device kitting addresses this directly by pre-assembling exactly what's needed for a procedure into a single, ready-to-use package.
This article covers what medical device kitting is, why it matters for both clinical outcomes and business operations, how the process works under strict regulatory oversight, what compliance demands you must meet, and how to find the right partner to manage it.
TLDR
- Medical device kitting bundles instruments, supplies, and components into ready-to-use kits that reduce surgical prep time by up to 8.3 minutes per procedure
- Benefits include 35% fewer procedural errors, inventory cost reductions up to 37.7%, and built-in regulatory compliance
- Assembly follows a defined workflow: assessment, procurement, controlled-environment build, quality control, and compliant distribution
- The right kitting partner should hold FDA/ISO credentials, offer kit customization, and connect directly with your existing supply chain
What Is Medical Device Kitting?
Medical device kitting is the process of assembling and packaging medical devices, instruments, supplies, and instructions into a single, ready-to-use kit. It's more than bundling products together. Kitting is a regulated manufacturing activity that falls under strict FDA requirements, quality management systems, and traceability standards.
The FDA explicitly classifies convenience kits as medical devices in their own right, meaning kit assemblers are subject to the same rigorous Quality Management System Regulation (QMSR) requirements as traditional manufacturers. Under 21 CFR 820.3, any entity that "assembles, processes, or packages a finished device" is considered a manufacturer and must comply with full quality system regulations.
What distinguishes medical device kitting from general product kitting:
- Validated sterile barrier systems that maintain product integrity until point of use
- Unique Device Identification (UDI) labels with production identifiers on every kit
- Full lot traceability for every component — from sourcing through final delivery — for recall readiness
- Controlled assembly environments with documented standard operating procedures
Medical device kits are used across a wide range of settings:
- Surgical suites, from routine procedures to complex operations
- Emergency response and disaster relief scenarios
- Clinical diagnostics and home care delivery
- Medical supply distribution, including e-commerce fulfillment and international shipping
Key Benefits of Medical Device Kitting
Streamlined Workflow and Time Savings
Pre-assembled kits eliminate the need for clinical staff to locate and gather individual components before procedures. Nurses spend about 6% of a 12-hour shift "hunting and gathering" equipment and supplies—time that comprehensive surgical packs can reclaim.
The efficiency gains are substantial. Studies on cataract and vitreoretinal surgery showed that comprehensive procedure packs reduced surgical preparation time by:
- 8.3 minutes per retina surgery in hospital settings
- 5.1 minutes per retina surgery in ambulatory surgery centers
- 7.9 minutes per cataract surgery in hospitals
If efficiently reallocated, these time savings could allow a hospital to perform over 250 additional cataract procedures per year.
Error Reduction and Standardization
Standardized kits ensure the same tools are used consistently across procedures and facilities, minimizing variability in care quality. A randomized controlled trial on central line catheter insertions demonstrated that prepackaged all-inclusive kits reduced:
- Major technical mistakes by 35%
- Minor technical mistakes by 35%
- Procedure duration by approximately 5 minutes

The prepackaged format reduces cognitive load on clinicians by eliminating the need to actively select additional items, which leads to fewer mechanical complications.
Inventory Optimization and Cost Savings
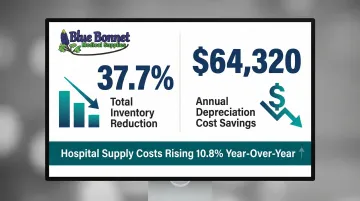
Kitting reduces overstocking and understocking risks by consolidating demand into predictable kit quantities. A study on perioperative inventory optimization demonstrated that standardizing and reducing tray sizes by 18% led to:
- Total inventory reduction of 37.7%
- $64,320 in annual depreciation cost savings
Reduced component-level inventory tracking also lowers carrying costs and shrinkage. With hospital supply expenses rising 10.8% year-over-year in early 2025, supply chain managers are under real pressure to find savings—and kitting is one of the few levers that cuts costs without cutting care quality.
Regulatory Compliance Built In
Properly designed kits come pre-labeled with lot numbers, expiration dates, sterility indicators, and usage instructions—making compliance documentation easier and reducing audit risk. Each kit label serves as a single traceability record, replacing the need to document dozens of individual components separately.
Key compliance elements built into well-designed kits include:
- Lot numbers and expiration dates on the outer label
- Sterility indicators visible before opening
- Usage instructions included inside each pack
- Manufacturer and regulatory markings for audit readiness
Competitive Advantage for Medical Product Businesses
For medical device manufacturers and online medical supply distributors, kitting creates a differentiated product offering that reduces per-order handling costs and speeds up fulfillment—particularly for Amazon or direct-to-consumer channels where pick accuracy matters.
Beyond logistics, kits ensure a manufacturer's product is used for a procedure as intended. That consistency protects both brand reputation and clinical outcomes.
Types of Medical Device Kitting Services
Custom Kitting
Custom kits are tailored to a specific healthcare provider's workflow, a particular surgical procedure, or a product manufacturer's unique configuration. This approach allows precise control over component selection, packaging format, and labeling requirements. Custom kitting is ideal when standardization across your organization or product line is essential, or when procedures have unique instrument requirements.
Standard Kitting
Where custom kits prioritize precision, standard kits prioritize volume. Pre-configured around commonly used component combinations, they're faster to produce and easier to stock — well-suited for high-volume distribution through e-commerce, wholesale, or multi-facility networks. Standard kits work best for routine procedures where clinical needs stay consistent across providers.
Key advantages of standard kitting:
- Faster turnaround from order to shipment
- Simpler inventory management for high-volume SKUs
- Lower per-unit cost compared to fully customized configurations
Emergency and Specialty Kitting
Emergency kits — built for trauma response, disaster relief, and field medicine — are designed around one principle: everything the responder needs must be immediately accessible without sorting through packaging. Incomplete or disorganized contents aren't just an inconvenience; in the field, they're a liability.
Specialty kitting covers a different but equally demanding set of requirements. Diagnostic and home-care kits are fast-growing categories that typically need:
- Consumer-friendly packaging designed for non-clinical users
- Multilingual labeling and instructions
- Compliance with FDA and applicable international regulations
The Medical Device Kitting Process, Step by Step
Assessment and Planning
The process begins by defining clinical or operational requirements: what components are needed, in what quantities, in what configuration, and for what end use. A kitting partner should resolve these upfront:
- What regulatory constraints apply (FDA, EU MDR, country-specific)?
- What sterility requirements must be met?
- What shelf life considerations affect component selection?
- What shipping environment will the kit encounter?
- What labeling requirements apply to your market?
Component Procurement and Inventory Management
Components are sourced from approved suppliers, verified against specifications, and logged into an inventory management system. Under 21 CFR 820.50, manufacturers must evaluate and select suppliers based on their ability to meet quality requirements — and purchasing documents must include agreements that suppliers will notify them of any changes.
Lot traceability starts here. Every component's lot number, expiration date, and supplier information must be captured and maintained all the way through final distribution.
Kit Design and Assembly
Design covers component arrangement for ease of use, packaging material selection (sterile barrier systems, tamper-evident seals), and label layout. Assembly must be performed in a controlled environment — cleanroom or controlled warehouse — by trained personnel following standard operating procedures.
Two standards govern this phase:
- ISO 14644-1:2015 — classifies air cleanliness by particle concentration, setting contamination control requirements for assembly environments
- ISO 11607-1 & 2:2019 — specifies requirements for materials, sterile barrier systems, and validation of forming, sealing, and assembly processes
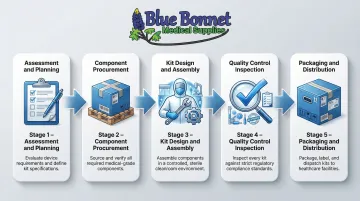
Quality Control Inspections
Before kits are sealed, inspections verify:
- Component completeness against the kit specification
- Absence of damage or expiration
- Label accuracy including UDI, lot numbers, and expiration dates
- Documentation of the inspection itself
Quality control records generated here become part of the kit's permanent documentation — traceable at every point from assembly through end use.
Packaging, Labeling, and Distribution
Finished kits are sealed, labeled with all required regulatory information, and prepared for storage or shipment. Distribution logistics — storage conditions, shipping methods, and chain-of-custody documentation — carry the same regulatory weight as the assembly process.
Temperature-sensitive kits require cold chain management. Sterile kits must be protected from any environmental exposure that could compromise barrier integrity during transit.
Compliance and Regulatory Requirements in Medical Device Kitting
FDA and International Regulatory Frameworks
In the U.S., medical device kitting must comply with FDA regulations including 21 CFR Part 820 Quality System Regulation and UDI labeling requirements. Effective February 2, 2026, the FDA's QMSR incorporates ISO 13485:2016 by reference, making this international standard the U.S. regulatory baseline.
Internationally, the EU Medical Device Regulation (MDR) 2017/745 governs systems and procedure packs under Article 22. The entity responsible for combining CE-marked devices into a procedure pack must assign a Basic UDI-DI to the pack and provide it to the EUDAMED database. Country-specific import regulations add additional layers of compliance.
Non-compliant kits can be rejected at customs or recalled, creating serious financial and reputational consequences for manufacturers and distributors alike. In 2025, Medline Industries recalled certain sterile convenience kits because they were mislabeled as sterile without completing the sterilization process, with FDA warnings that use could cause bloodstream infections, sepsis, and death.
Sterility Assurance Requirements
Many medical device kits require sterile packaging validated through established sterilization protocols. Packaging must be validated to maintain sterility until the point of use. Any breach in sterile barrier integrity from improper packaging, handling, or shipping renders a kit non-compliant and potentially unsafe.
Traceability and Documentation Obligations
Sterility requirements don't stand alone — traceability obligations run in parallel, covering the full chain from component sourcing to final delivery. Required documentation includes:
- Lot numbers and expiration dates for all components
- Assembly records and quality control results
- Distribution logs tracking each kit's movement
- Complaint records covering device, labeling, or packaging failures
This documentation is the backbone of any recall response, adverse event investigation, or regulatory audit.
Working with a Compliant Fulfillment Partner
Given these documentation and traceability demands, your fulfillment partner's credentials matter as much as your own. Look for FDA registration, ISO 13485 or GMP compliance, and HIPAA-safe handling practices backed by a documented quality management system.
Bluebonnet Medical Supplies holds FDA clearance for medical packaging services and operates under ISO and GMP standards — giving medical product businesses compliant storage, packing, and shipping without the overhead of building that infrastructure themselves.
How to Choose the Right Medical Device Kitting Partner
Evaluate Compliance Credentials and Quality Systems First
A partner's certifications—FDA, ISO, GMP—should be verified, not just claimed. Ask for documentation, audit history, and how they handle deviations or non-conformances. Request evidence of:
- Current ISO 13485 certification with documented processes mapping to FDA QMSR requirements
- Validated sterile barrier systems with process qualification documentation
- Cleanroom controls with continuous environmental monitoring logs
- UDI labeling capability and experience with GUDID submissions

Any partner who can't answer these questions with specifics warrants a closer look before signing anything.
Assess Customization Capability and Supply Chain Integration
The right partner can adapt to your specific kit configuration, labeling requirements, and fulfillment channel—whether you're shipping to hospitals, fulfilling Amazon orders, or exporting internationally.
Bluebonnet Medical Supplies operates on exactly this principle. As a personal-touch 3PL, they build custom logistics solutions around each client's workflow, with kitting services that span FDA-cleared medical packaging to custom assembly across diverse distribution channels.
Consider Scalability, Communication, and Ongoing Support
Kitting needs shift as your product line grows, so evaluate partners on more than their current capabilities. Look for:
- Ability to scale volume up or down without service disruption
- Integration with your inventory management systems
- Returns handling and product restoration services
- Transparent, proactive communication (not just incident response)
A partner who treats your business individually—not as another account number—reduces operational risk over the long term.
Frequently Asked Questions
What is medical kitting?
Medical kitting is the process of assembling medical devices, supplies, and instructions into a single, ready-to-use package designed to improve clinical workflow efficiency, reduce errors, and ensure all necessary components are available at the point of care.
How much does a medical care kit cost?
Costs vary widely depending on the components included, level of customization, sterility requirements, volume, and complexity of compliance documentation. Request a quote from a kitting provider based on your specific configuration and order volume for accurate pricing.
What is the difference between standard and custom medical kitting?
Standard kits are pre-configured for common clinical applications and produced at volume. Custom kits are tailored to a specific provider's workflow, procedure, or product configuration. They offer greater precision but typically require more upfront planning.
What regulations apply to medical device kitting?
Key U.S. requirements include FDA 21 CFR Part 820 (Quality System Regulation) and UDI labeling. International standards include ISO 13485 and EU MDR 2017/745. Sterilization validation, traceability documentation, and proper labeling are universal compliance requirements across all markets.
Can medical device kits be shipped internationally?
Yes, but international shipping requires compliance with destination-country import regulations, correct documentation (lot numbers, labeling in local language, country-specific certifications), and a logistics partner experienced in customs clearance for regulated medical products.
Who typically handles medical device kitting?
Kitting is handled either in-house by manufacturers or outsourced to third-party contract packagers or 3PL providers with medical-grade facilities and compliance credentials. Providers like Bluebonnet Medical Supplies operate FDA-cleared, ISO and GMP-compliant facilities, making outsourcing a practical option for businesses that want to focus on growth rather than logistics.


