
Introduction
In medical supply and distribution, a compromised package doesn't just create a business problem — it puts patients at risk. Pharmaceutical recalls cost between $10 million and $100 million on average, with sterility failures accounting for roughly 38% of all drug recalls. The regulatory and reputational fallout can take years to recover from.
Tamper-evident packaging is the first line of protection, but no single solution fits every situation. The right choice depends on your product type, distribution channel, and regulatory requirements. Consider what's actually at stake:
- A liquid medication needs a hermetic seal that blocks contamination
- A sterile surgical device requires packaging that holds sterility through transport
- A controlled substance demands documented chain-of-custody protection
This article walks through 10 types of tamper-evident packaging, how each one works, and what to consider when choosing the right option for your medical products.
TLDR
- Tamper-evident packaging provides visible proof if a product has been opened or altered before reaching the end user
- FDA-mandated for OTC drugs under 21 CFR 211.132, and standard practice across pharmaceuticals, medical devices, and supplements
- Ten main types covered here include induction seals, blister packs, shrink bands, shrink wrap, security bags, tear-away labels, breakaway caps, security tape, heat-sealed pouches, and child-resistant packaging
- Selection depends on product format, regulatory requirements, distribution channel, and tamper resistance needed
- When choosing a 3PL, verify they apply tamper-evident packaging consistently and can document compliance with FDA and GMP standards
What Is Tamper-Evident Packaging?
Tamper-evident packaging is any packaging that includes one or more indicators or barriers to entry that — if broken or missing — provide visible evidence to the consumer that tampering has occurred. The FDA formally defines this under 21 CFR 211.132, requiring that such packaging be "distinctive by design" and employ identifying characteristics that cannot be duplicated with commonly available materials.
The key distinction: tamper-evident packaging does not necessarily prevent tampering — it makes tampering visible. Tamper-resistant packaging, by contrast, is designed to make access more difficult in the first place. Most effective solutions combine both approaches, creating barriers that are hard to bypass while leaving unmistakable evidence if someone tries.
To qualify as tamper-evident, packaging must create actual evidence — visible damage, missing components, or altered appearance. Solutions that can be removed and reapplied without a trace do not meet the standard. That requirement applies across every regulated category: food, pharmaceuticals, medical devices, cosmetics, and dietary supplements.
Why Tamper-Evident Packaging Matters in Medical Supply and Distribution
Regulatory Stakes Are High
The FDA mandates tamper-evident packaging for over-the-counter drug products, and medical device packaging must meet standards demonstrating that sterility and integrity have been maintained. ISO 11607 governs sterile medical device packaging, specifying requirements for materials, sterile barrier systems, and validation processes. Non-compliance can lead to recalls, customs holds, or regulatory action — a real risk for businesses selling across borders.
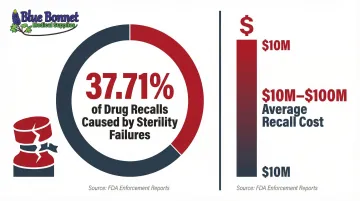
The consequences of packaging failures are severe. Sterility issues account for approximately 37.71% of drug recalls, and recalls cost between $10 million and $100 million on average. The FDA has issued warnings about eye drop bottles with loose safety seals that fall off during use, presenting contamination hazards. Medical devices like surgical mesh have faced Class II recalls due to foreign contaminants found inside primary sterile packaging.
What Goes Wrong Without It
Without proper tamper-evident packaging:
- Product contamination — Compromised seals allow bacteria, moisture, or foreign materials to enter
- Liability exposure — If a patient is harmed by a tampered product, legal and financial consequences follow
- Loss of customer trust — Once consumers question product safety, rebuilding confidence takes years
- Reputational damage — Recalls and safety incidents generate negative publicity that impacts future sales
Protection Throughout the Distribution Chain
Tamper-evident packaging matters not just at the point of manufacture, but at every stage — storage, picking, packing, and last-mile delivery. A product can leave the production line perfectly sealed and still arrive compromised if handling downstream isn't held to the same standard.
For medical product businesses using a third-party logistics provider, that's where the operational details matter. Bluebonnet Medical Supplies handles FDA-compliant packaging and GMP-compliant packing processes as part of its standard warehouse operations — including documentation, labeling, and proper tamper-evident application before products ship domestically or internationally.
10 Types of Tamper-Evident Packaging and How They Work
Not all tamper-evident packaging works the same way. Some rely on heat bonding, others on mechanical breaks or adhesive chemistry. The right choice depends on your product type, regulatory requirements, and distribution environment. Here's how each method works and where it fits.
Induction Seals
Induction seals use electromagnetic induction heat to bond a foil liner inside a cap to the bottle or container opening. The process melts a polymer coating to form a hermetic seal that must be visibly punctured or peeled to access the product. This is one of the most common forms of tamper evidence for bottled liquids in pharmaceuticals and supplements.
Common applications:
- Liquid medications requiring airtight seals
- Nutritional supplements and vitamins
- Bottled medical or chemical products
- Products with high shelf-life requirements
- International shipments where contamination prevention is critical
The FDA's Compliance Policy Guide notes that induction seals offer a "higher degree of tamper-resistance" than adhesive seals, making them ideal for high-risk liquid products.
Shrink Bands
Shrink bands are plastic sleeves applied around the cap and neck of a container, then heat-shrunk to conform tightly. They must be torn or cut to open the cap, leaving clear evidence of access. While PVC currently holds 32.4% of the shrink band market, the industry is shifting toward recyclable polyolefin materials to support closed-loop recycling.
Typical uses:
- Bottles, jars, and containers in pharma and personal care
- Single-serve items where shelf integrity matters
- Retail products requiring visible consumer protection
- Products needing cost-effective tamper evidence
Under FDA requirements, shrink bands must employ an identifying characteristic — tinted bands alone no longer meet the standard.
Blister Packs
Blister packs consist of pre-formed plastic cavities sealed with foil or plastic backing. Each cavity holds an individual unit (tablet, capsule, device component), and accessing a unit requires visibly puncturing the backing — making it immediately obvious which units have been used or tampered with.
Where it fits:
- Unit-dose pharmaceuticals and OTC medications
- Small medical device components
- Hospital and clinical settings where dose accountability is essential
- Products requiring individual unit protection
The FDA explicitly recognizes blister packs as tamper-evident packaging, noting that the backing cannot be separated without visible evidence of entry.

Lidding Films
Lidding films are thin laminate films applied to the top of trays, cups, or containers using heat sealing. They are designed with a pull tab for easy opening, but once peeled back, they cannot be resealed — leaving visible evidence of access.
Ideal for:
- Medical trays and diagnostic kits
- Sterile specimen containers
- Laboratory supply products shipped to healthcare settings
- Food and dairy products requiring single-use protection
Tamper-Evident Labels and VOID Labels
Tamper-evident labels are pressure-sensitive adhesive labels designed to leave behind a visible mark (such as the word "VOID" or a residue pattern) if removed from the package. Standard labels do not meet this standard — the key is whether removal creates irreversible evidence.
Used primarily for:
- Cartons, boxes, and secondary packaging as a supplementary seal
- Bottles or pouches as a cost-effective layer of protection
- Pharmaceutical supply chain cartons requiring visual verification
Labels alone are often insufficient for high-risk products and work best paired with a mechanical or heat-bonded method.
Security Tape
Security tape is specialized adhesive tape engineered to display "VOID," "OPENED," or a pattern when removed from a surface, making it impossible to reseal a box or carton without evidence. It is applied over carton seams or closures during packing.
Common applications:
- Shipping cartons and outer boxes
- Transport packaging for medical devices and supplies
- Distribution chain verification — shows whether packages were opened in transit
- Secondary packaging requiring documented integrity
Breakaway Caps and Ratchet Closures
Breakaway caps are bottle closures with a perforated ring or ratchet mechanism that disconnects from the cap when first opened, leaving a broken ring visibly attached to the bottle neck. These are mechanical rather than adhesive — the physical structure itself is the evidence.
Typical uses:
- Bottled liquids such as medications and topical solutions
- Medical-grade chemicals
- Over-the-counter and prescription products
- Applications requiring quick, intuitive tamper verification at point of use
Resealable Pouches with Tear Notches
Resealable pouches with tear indicators require tearing a notch or perforation above the zipper to first open. Once the tear is made, it is permanently visible — even if the pouch is resealed with the zipper, the tear cannot be hidden.
Ideal for:
- Powdered supplements
- Dehydrated or bagged medical consumables
- Wound care accessories
- Flexible-format medical supplies requiring both tamper evidence and consumer convenience
Heat-Sealed Pouches
Heat-sealed pouches have all edges fused through heat and pressure, creating a completely sealed enclosure that must be torn or cut open. These are distinct from resealable pouches — they are intended as single-use, and any prior opening is immediately visible.
Where it fits:
- Sterile medical devices and surgical instruments
- Dressings and implants
- Products requiring sterile barrier packaging
- Applications aligning with ISO 11607 requirements for maintaining sterility through point of use
Most heat-sealed pouches combine a clear film layer with a porous material — like Tyvek or medical-grade paper — so sterilant gas can penetrate before sealing.
Tamper-Evident Security Bags
Tamper-evident security bags are specialized poly or paper bags with a self-sealing closure that permanently bonds on first use. Attempting to open and reseal the bag leaves visible signs — tearing, deformation, or a "VOID" indicator along the seal strip.
Used primarily for:
- Evidence packaging and chain-of-custody documentation
- High-value medical devices
- Controlled substances requiring DEA compliance under 21 CFR 1302.06
- Sensitive medical products during packing and outbound shipping
Fulfillment centers handling medical products — including 3PL providers like Bluebonnet Medical Supplies — use security bags to maintain documented chain-of-custody through every stage of outbound shipping.
How to Choose the Right Type — and Mistakes to Avoid
Four Key Variables
The right tamper-evident solution depends on:
- Product format — Liquid, solid, device, or sterile product
- Distribution channel — Retail, direct-to-consumer, clinical, or international
- Regulatory requirements — FDA OTC rules, ISO 11607, customs requirements
- Level of evidence needed — Primary product protection vs. secondary packaging security
Rule of thumb: Products where tampering could harm users need the highest-integrity solutions — induction seals, heat-sealed pouches, blister packs. Secondary packaging in the supply chain benefits from security tape and tamper-evident bags.
Common Selection Mistakes
Choosing complexity over fit:
- Selecting the most expensive option when a simpler one meets the use case
- Relying on a single method when layering two types provides stronger protection
- Choosing based on familiarity rather than actual product and regulatory fit
Underestimating application consistency:
- Even the best tamper-evident format fails if applied inconsistently during packing
- Lack of documented procedures leads to gaps in protection
- Insufficient training results in improper seal application
Ignoring secondary packaging:
- Focusing only on primary product packaging while neglecting outer cartons
- Missing opportunities to add security tape or bags during distribution
- Failing to protect products during transit and warehousing

The 3PL Factor
Selecting the right packaging format only gets you halfway there — how consistently it's applied during fulfillment is equally important. If you're working with a 3PL partner, confirm that tamper-evident packaging is part of a documented, compliant packing process, not an afterthought.
Bluebonnet Medical Supplies handles fulfillment under FDA, ISO, and GMP compliance standards, so tamper-evident requirements are built directly into the packing workflow for every order.
Frequently Asked Questions
What is tamper-evident packaging?
Packaging with one or more indicators or barriers to entry that, if breached or missing, provide visible evidence that tampering has occurred. Under FDA definition 21 CFR 211.132, it must be distinctive by design and cannot be duplicated with commonly available materials. It shows evidence rather than prevents access outright.
What is the purpose of tamper-evident packaging?
It protects consumers from receiving compromised products, deters deliberate tampering, helps businesses meet regulatory requirements, and reduces liability risk — especially in food, pharmaceuticals, and medical devices where contamination or alteration poses direct health risks.
What are four types of tamper-evident packaging?
The most widely used types are induction seals (for liquid bottles), blister packs (for unit-dose tablets), shrink bands (for jars and containers), and heat-sealed pouches (for sterile devices). Each is suited to different product formats and industries.
What is tamper-proof packaging?
Tamper-proof packaging is designed to physically prevent or significantly resist unauthorized access, not just show evidence of it. True tamper-proofing is difficult to achieve, so most packaging combines tamper-resistant features (making access difficult) with tamper-evident features (showing when access occurred).
Is tamper-evident packaging required by law for medical products?
The FDA requires tamper-evident packaging for over-the-counter drug products under 21 CFR 211.132. Sterile medical device packaging must meet ISO 11607 standards. Requirements vary by product classification, so businesses should verify which rules apply to their specific product category and distribution channel.
Can one type of tamper-evident packaging be enough, or do you need multiple?
One method may meet minimum regulatory requirements, but industry best practice in medical and pharmaceutical applications involves layering two or more tamper-evident features. For example, an induction seal on the bottle plus security tape on the outer carton covers primary, secondary, and transit packaging for comprehensive protection.


