
Introduction
A single medical device recall can cost a manufacturer anywhere from $10 million to $600 million — and 49% of that financial damage comes from business disruption and lost sales alone. Most of those situations trace back to packaging failures: wrong materials, missed standards, or a compliance gap that slipped through.
Medical-grade plastic packaging directly affects patient safety, product integrity, and regulatory standing. Get it wrong and you're looking at rejected shipments, penalty exposure, or compromised products reaching end users.
This guide covers what medical-grade plastic is, why it's different from ordinary plastic, the key standards businesses must know (ISO, FDA, USP), and what to look for in a compliant packaging and fulfillment setup.
TLDR
- Medical-grade plastic is engineered for biocompatibility—it won't react with the human body or the product it contains
- Compliance is determined by standards like ISO 10993, USP Class VI, and FDA guidelines, not a single universal definition
- Blister packs, sterile pouches, vials, trays, and bottles are the most common medical-grade packaging formats
- Non-compliance can result in product recalls, customs holds, or liability exposure
- Partnering with a regulatory-compliant 3PL ensures your packaging meets FDA, ISO, and GMP requirements without managing it in-house
What Is Medical-Grade Plastic?
There is no single regulatory definition of "medical-grade plastic." Instead, a plastic is considered medical-grade when it meets recognized standards for biocompatibility and safety in medical applications, as outlined by bodies like ISO and the U.S. Pharmacopeial Convention (USP).
Biocompatibility means the plastic must be safe for direct or indirect contact with the human body. According to the FDA, biocompatibility is "the ability of a device material to perform with an appropriate host response in a specific situation."
That means the material must be non-toxic, non-reactive, and chemically stable under real-world conditions—exposure to bodily fluids, sterilization processes, and extended storage.
Core properties that differentiate medical-grade plastics from standard industrial plastics include:
- Resistance to temperature extremes
- Chemical inertness
- Ability to withstand sterilization cycles (gamma radiation, ethylene oxide, autoclaving)
- Long-term stability without leaching harmful substances
These requirements apply more broadly than many assume. Even single-use medical packaging—pouches, blister packs—must meet medical-grade standards. Regulators evaluate the entire system (material + packaging + manufacturing process), not just reusable devices.
How It Differs from Regular Plastic
While everyday plastics (like food-grade PE or consumer PP) may look identical, they are formulated differently. Medical-grade resins go through stricter purity controls, with tightly controlled additives, colorants, and processing aids to minimize the risk of leachable contaminants.
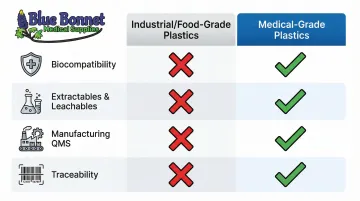
| Control Area | Industrial/Food-Grade Plastics | Medical-Grade Plastics |
|---|---|---|
| Biocompatibility | Not required; food-contact limits apply | Must pass ISO 10993 or USP testing |
| Extractables & Leachables | Basic migration limits | Rigorous E&L profiling and toxicological risk assessments |
| Manufacturing QMS | Standard ISO 9001 or GMP for food | Must comply with 21 CFR Part 820 and/or ISO 13485 |
| Traceability | Lot-level tracking | Strict batch/lot traceability; UDI integration on packaging |
The manufacturing environment matters: medical-grade plastics are typically produced under GMP (Good Manufacturing Practice) conditions, with batch traceability—something standard plastic manufacturing does not require.
Common Types of Medical-Grade Plastic and Packaging Formats
Types of Medical-Grade Plastics Used in Packaging
Five polymers dominate medical packaging — each with distinct strengths, sterilization limits, and compliance considerations:
- PE (Polyethylene): Flexible films, bags, and bottles. Handles gamma and EtO well; not suited for autoclave.
- PP (Polypropylene): Rigid autoclavable containers. Vulnerable to chain scission under gamma radiation unless radiation-stabilized grades are specified.
- PETG: Blister trays and clamshells. Its amorphous structure enables consistent thermoforming and maintains transparency post-sterilization — useful when visual product inspection is required.
- PVC (Flexible): Still common in blister and pouch formats, but declining. The FDA and EU REACH Annex XVII (Entry 51) restrict DEHP and other ortho-phthalate plasticizers due to developmental and reproductive toxicity risks.
- ABS and PEEK: ABS suits rigid protective cases with good radiation resistance. PEEK handles up to 3,000 autoclave cycles at 134°C without hydrolysis or mechanical degradation — the standard choice for reusable sterilization trays.
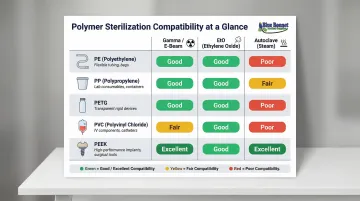
Polymer Compatibility Matrix:
| Polymer | Primary Applications | Sterilization Compatibility | Regulatory Notes |
|---|---|---|---|
| PE (HDPE/LDPE) | Films, bags, pouches, bottles | Good for Gamma/E-beam and EtO; Poor for Autoclave | Meets USP standards; highly stable |
| PP | Thermoformed trays, autoclavable containers | Good for Autoclave and EtO; Poor for Gamma (unless stabilized) | Susceptible to chain scission under radiation |
| PETG | Rigid thermoformed trays, blisters | Good for Gamma, E-beam, and EtO; Poor for Autoclave | Maintains clarity post-sterilization |
| PVC (Flexible) | Pouches, bags, tubing | Good for Gamma, E-beam, and EtO; Fair for Autoclave | High regulatory scrutiny regarding phthalate leaching |
| PEEK | Reusable sterilization trays, lids | Excellent for Autoclave, Gamma, and EtO | Withstands 3,000+ autoclave cycles |
Packaging Formats for Medical Products
The right format depends on your product type, sterilization method, and distribution channel. Three formats cover the majority of medical packaging applications:
Sterile pouches (Tyvek/plastic composite): The default choice for surgical instruments and single-use devices. A porous Tyvek substrate heat-sealed to a transparent film provides a validated microbial barrier, clean-peel opening, and compatibility across EtO, Gamma, E-beam, and controlled steam sterilization cycles.
Blister packs and trays: Used for pharmaceuticals and small devices. PETG is the most common forming material — its transparency allows visual inspection, and validated form/fill/seal integrity keeps products sterile through distribution and storage.
Bottles, vials, and ampoules: Standard for liquid pharmaceuticals and reagents. Closure systems (caps, septa) must use inert, compatible materials that won't react with the product or degrade the seal across the labeled shelf life.
Regulatory Standards That Govern Medical-Grade Plastic Packaging
Compliance is multi-layered: material-level testing, device/packaging-level testing, and process-level compliance (GMP) all interact. No single certification covers everything.
ISO 10993: The Biocompatibility Standard
ISO 10993 is the primary international standard for evaluating the biocompatibility of medical device materials, including packaging that contacts the product. It requires biological evaluation covering:
- Cytotoxicity
- Sensitization
- Irritation
- Systemic toxicity
ISO 10993 also encompasses risk management analysis of manufacturing processes, additives, and degradation products. The FDA now explicitly prefers the risk-based, in vitro approach of the ISO 10993 series over traditional animal testing, with particular emphasis on ISO 10993-18 chemical characterization.
USP Class VI
USP Class VI is the most rigorous of the six USP plastic classifications. It involves systemic injection, intracutaneous, and implantation tests in live animal models to evaluate toxicity.
USP Class VI is widely recognized by the FDA, but it does not include the risk management component of ISO 10993. The two standards have different scopes — passing one does not automatically confer compliance with the other.
The USP utilizes a dual-chapter framework to evaluate plastic packaging:
- USP <661.1>: Tests raw polymers for identity, biocompatibility, physicochemical properties, and composition
- USP <661.2>: Evaluates the finished, assembled packaging system through chemical and toxicological testing to confirm safety for end use
FDA Requirements for Medical Packaging in the U.S.
The FDA does not "approve" specific plastics. Rather, it clears entire medical devices (including their packaging) under 510(k) or PMA pathways. For packaging specifically, FDA guidance requires that packaging maintains sterile barrier integrity, protects the device through its labeled shelf life, and does not introduce contamination.
These clearance requirements translate into specific regulatory obligations at the facility and process level:
- FDA 21 CFR Part 820 (Quality System Regulation / GMP): Governs facility standards, documentation, traceability, and process controls
- 21 CFR 820.45: Mandates that manufacturers document and maintain procedures to ensure packaging integrity during processing, storage, handling, and distribution
- 21 CFR 820.130: Covers device packaging and labeling controls
GMP compliance is required for businesses manufacturing or contract-packing medical products. It extends to whoever handles, packs, and ships those products.
ISO 11607: Sterile Barrier Validation
ISO 11607 governs sterile barrier systems:
- ISO 11607-1: Specifies requirements and test methods for materials, preformed sterile barrier systems, and packaging systems intended to maintain sterility until the point of use
- ISO 11607-2: Specifies validation requirements for processes used to form, seal, and assemble sterile barrier systems
Manufacturers must establish appropriate upper and lower processing limits (temperature, pressure, dwell time) and demonstrate process reproducibility through Installation Qualification (IQ), Operational Qualification (OQ), and Performance Qualification (PQ).
ASTM Test Methods for Sterile Barrier Validation:
| ASTM Standard | Test Description | Purpose |
|---|---|---|
| ASTM F88 | Seal Strength of Flexible Barrier Materials | Quantifies mechanical force required to separate a seal |
| ASTM F1929 | Dye Penetration for Porous Packaging | Detects micro-channels or pinhole leaks down to 50 µm |
| ASTM F2096 | Internal Pressurization (Bubble Test) | Detects gross leaks by observing escaping bubbles |
| ASTM F1980 | Accelerated Aging of Sterile Barrier Systems | Simulates time effects to establish expiration dates |

Why Packaging Compliance Matters for Your Business
The direct business risks of non-compliant packaging are severe:
- Customs seizures and holds for international shipments
- Product recalls costing $10–600 million per event
- Regulatory warning letters from the FDA
- Liability exposure if a product reaches an end user in compromised condition
Consider what that looks like in practice: Medtronic recalled non-absorbable sutures after manufacturing errors created gaps or wrinkles in the breather pouch seal, breaching the sterile barrier and creating infection risk. That's a FDA Class I recall — the category reserved for products with a reasonable probability of causing serious injury or death.
Platform and Partner Trust
In the medical supply space, one packaging failure can damage relationships with distributors, healthcare providers, or platform partners. Amazon requires sellers of Class I and II non-exempt medical devices to provide proof of FDA facility registration and 510(k) clearance. Products must also be sold in "New" condition within original, unopened, and properly sealed packaging. Amazon prohibits products subject to unresolved FDA enforcement.
Cross-Border Compliance
Different markets have different requirements — EU MDR, FDA, and ISO standards don't always align. Packaging that passes in the US may need modification before it ships internationally. Under EU MDR 2017/745 Annex I (GSPR), sterile devices must remain sterile through all transport and storage conditions. That obligation doesn't end at the factory door; it extends to how the product is packed and labeled for its entire journey.
Choosing the Right Medical Packaging and Fulfillment Partner
The key credentials to look for in any partner who will touch your medical products:
- FDA-cleared packaging operations
- ISO compliance
- GMP compliance
- HIPAA-safe handling practices
These are non-negotiable—your brand carries the liability, even if a third party handles fulfillment.
What compliant warehousing and packing looks like:
- Controlled storage environments
- Trained staff
- Proper documentation and lot traceability
- Validated pack processes
- Ability to handle returns without compromising product integrity
Under 21 CFR 820.3, the definition of a "Manufacturer" includes third parties performing contract sterilization, repacking, or relabeling. This means contract packagers are subject to FDA inspection and must maintain the same QMS standards as device manufacturers.
That regulatory exposure is exactly why choosing the right 3PL partner matters. Bluebonnet Medical Supplies holds FDA-cleared medical packaging services, ISO, and GMP compliance, with customized solutions built around each client's specific workflow—not a one-size-fits-all model.
Their core services include:
- Documentation, labeling, and lot traceability integrated into daily operations
- Inventory tracking and validated handling procedures
- International shipping support to keep products clear of customs delays
- Dedicated Amazon FBA medical supply fulfillment with built-in compliance checks

Frequently Asked Questions
What is medical grade plastic called?
Medical-grade plastic is commonly called a "medical-grade polymer" or "biocompatible plastic." The designation means the material meets biocompatibility and safety standards—such as ISO 10993 or USP Class VI—required for use in medical devices and packaging.
What is medication packaging called?
Medication packaging is broadly referred to as "pharmaceutical packaging" or "primary packaging." Common specific formats include blister packs, ampoules, vials, and bottles. The term "sterile barrier system" is used when the packaging must maintain sterility for medical devices.
What standards must medical-grade plastic packaging meet?
Key standards include ISO 10993 for biocompatibility, USP Class VI for toxicological testing, ISO 11607 for sterile barrier systems, and FDA guidelines (including 21 CFR Part 820 for GMP) in the U.S. market. Requirements vary by product type and target market.
Is all plastic packaging safe for medical devices?
No—standard commercial or food-grade plastics are not automatically safe for medical devices. Medical-grade plastics are specifically formulated and tested to ensure they don't leach harmful substances, react with the product, or degrade under sterilization conditions.
What happens if medical packaging doesn't meet compliance standards?
Non-compliant packaging can trigger product recalls costing $10–600 million, regulatory enforcement actions, customs seizures for international shipments, and liability for patient harm. Platforms like Amazon may also delist non-compliant medical products.
Do medical products need special storage conditions between packaging and shipping?
Many medical products require controlled storage environments—specific temperature ranges, humidity controls, or protection from light—to maintain packaging integrity and product stability. GMP-compliant warehouses are designed to meet these requirements and document conditions throughout storage.


