
Introduction
Medical flexible packaging does far more than wrap a product: it's the first line of defense for sterility, patient safety, and regulatory compliance. Getting it wrong can cost businesses time, money, and market access. According to FDA data, packaging and labeling issues account for 13% of all medical device recalls, with a single major recall costing up to $600 million when you factor in product disposal, logistics disruption, and litigation.
This guide is for medical product sellers, device distributors, and online healthcare retailers who need to understand packaging requirements before their products ever leave the warehouse.
Whether you're shipping surgical instruments, wound care supplies, or diagnostic devices, your packaging choice determines whether products arrive sterile, pass customs clearance, and meet FDA requirements—or face costly compliance failures.
TLDR
- Medical flexible packaging uses PE films, laminates, and Tyvek® to maintain sterile barriers during storage and transport
- Common formats include pouches, blister packs, IV bags, FFS films, and thermoformed trays — each matched to specific sterilization methods
- FDA clearance, ISO 11607, and GMP compliance are non-negotiable for US and international distribution
- Packaging decisions directly affect shipping costs, customs clearance, and product integrity on arrival
- Poor material selection is one of the most preventable causes of recalls and regulatory delays
What Is Medical Flexible Packaging?
Medical flexible packaging refers to packaging made from non-rigid materials—films, foils, laminates, and nonwovens—specifically engineered to maintain sterile barriers and protect healthcare products throughout storage, transport, and use.
Unlike rigid formats like glass vials or hard plastic containers, flexible packaging is lighter, adapts to product shape more easily, and typically costs less per unit—without sacrificing sterility or safety standards.
The regulatory definition that matters most is the "sterile barrier system" (SBS). According to ISO 11607-1:2019, an SBS is the minimum packaging required to allow sterilization, provide a microbial barrier, and allow for aseptic presentation. This definition separates the sterile barrier from protective packaging, which simply protects the SBS during distribution.
Where medical flexible packaging is used:
- Hospital surgical centers receiving sterile instruments and implants
- Pharmaceutical supply chains distributing unit-dose medications
- Home healthcare product shipments for patient use
- E-commerce fulfillment for medical supplies sold online
- International distribution requiring customs documentation
Get the packaging wrong, and products that passed every quality check in your facility can fail at the border or arrive compromised at the point of care.
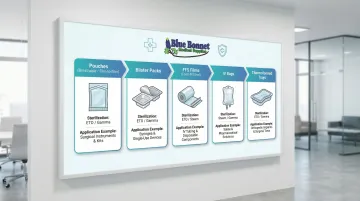
Common Types of Medical Flexible Packaging
Pouches (Breathable and Film-to-Film)
Breathable pouches are engineered for Ethylene Oxide (ETO) sterilization, using materials like Tyvek® or medical-grade paper that allow gas exchange while maintaining a microbial barrier. Film-to-film pouches suit gamma radiation or e-beam sterilization and offer stronger puncture resistance for heavier devices like surgical instruments and catheters.
Key applications: Catheters, syringes, wound dressings, surgical kits
Blister Packaging
Blister packs use a formed plastic cavity (PVC, PET, or PP) heat-sealed to a foil or paper backing to individually contain tablets, capsules, or small devices. Blister packaging accounts for 58% of the pharmaceutical unit-dose packaging market and over 70% of solid oral dosage packaging in developed markets due to superior tamper-evidence and dose-accuracy benefits.
Key applications: Tablets, capsules, transdermal patches, small diagnostic devices
Form Fill Seal (FFS) Films
FFS packaging is produced from a roll of flat film that is simultaneously formed, filled, and sealed in automated equipment. At high volumes, the per-unit cost drops significantly. Consistent packaging dimensions also reduce shipping costs — a practical advantage for wound care products, gloves, and surgical kits.
Common applications include:
- Wound care products and sterile gauze
- Surgical drapes and gloves
- High-volume disposable device packaging
IV Bags and Flexible Fluid Containers
IV bags represent one of the most regulated flexible packaging formats—typically made from PVC or non-PVC polyolefin films. Each bag must hold up chemically against its drug contents, stay sterile through aseptic filling, and maintain integrity across an extended shelf life. The EU REACH regulation mandates authorization for DEHP in medical devices by 2029, forcing a rapid shift to non-PVC alternatives.
Key applications: Intravenous solutions, irrigation fluids, parenteral nutrition
Thermoformed Trays with Lidding
Thermoformed trays are custom-shaped rigid-flex hybrids where a cavity is molded to the device's exact form and sealed with a breathable or foil lid. They protect high-value instruments (orthopedic implants, cardiac stents) and ensure devices reach the operating room undamaged and sterile.
Key applications: Orthopedic implants, cardiac stents, surgical instrument sets
Key Materials Used in Medical Flexible Packaging
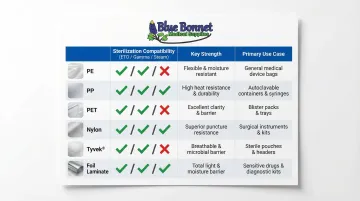
Polymer Films (PE, PP, PET, Nylon)
Each material serves a specific functional role:
- PE (Polyethylene): Forms heat-sealable, moisture-resistant layers that hold up well under gamma and e-beam sterilization
- PP (Polypropylene): Handles chemical exposure and autoclave conditions (121°C steam) well, but embrittles under gamma radiation above 20–50 kGy — a key limitation to plan around
- PET (Polyethylene Terephthalate): Structural strength and clarity when laminated
- Nylon (Polyamide): Puncture resistance for demanding applications
DuPont™ Tyvek® and Medical-Grade Paper
Tyvek® is a nonwoven HDPE material with a strong microbial barrier, good tear resistance, and broad sterilization compatibility — ETO, gamma, and steam all work with it. Tyvek 1073B targets high-risk devices with superior puncture resistance and maintains sterility for at least five years. Tyvek 1059B covers medium-risk items at a lower cost with the same sterility shelf life.
Medical-grade paper fills a similar role in lower-cost pouches, meeting sterilization breathability requirements where Tyvek's performance level isn't required.
Multi-Layer Laminates and Foil
High-barrier laminates combine multiple material layers (for example, PET/foil/PE) to block oxygen, moisture, and light — protection that's critical for drug-coated implants, diagnostics, and anything sensitive to environmental degradation.
Pharmaceutical blister packs are a practical example: aluminum foil backing provides near-impermeable protection in hot, humid climates where single-layer films would fall short.
Benefits of Flexible Over Rigid Healthcare Packaging
Protection Without Excess Bulk
Flexible packaging conforms to product shape, reducing interior movement and the risk of damage during shipping while adding far less weight and dimensional volume than rigid alternatives. This directly lowers freight costs per unit, especially under carrier DIM weight pricing.
Sterilization Compatibility and Seal Integrity
Properly engineered flexible packages maintain a sterile barrier through validated sterilization methods and across the full distribution chain. Key handling advantages include:
- Peel-open designs that allow contamination-free access at the point of care
- Consistent seal integrity across EO, gamma, and steam sterilization cycles
- Aseptic presentation that meets point-of-care requirements without secondary prep
Sustainability and Reduced Material Waste
Sustainability and Reduced Material Waste
Beyond sterility, packaging waste is a growing pressure point. US healthcare generates 1.7 million tons of plastic waste annually, yet less than 5% of healthcare plastics are currently recycled. Mono-material and reduced-layer flexible formats generate less landfill waste than multi-component rigid cartons — a concrete advantage as health systems and major retailers tighten their sustainability sourcing requirements.

Regulatory Compliance for Medical Flexible Packaging
FDA Requirements Overview
The FDA regulates medical device packaging under 21 CFR Part 820 (Quality System Regulation). Packaging must be part of the device's Design History File (DHF), with documented validation testing that covers seal integrity, sterile barrier maintenance, aging, and distribution simulation—not just material choice.
The new Quality Management System Regulation (QMSR) effective February 2, 2026 aligns 21 CFR Part 820 with ISO 13485:2016. Under the new §820.45, manufacturers face stricter mandates to document the inspection of labeling and packaging prior to release to prevent mix-ups.
ISO 11607 as the Global Standard
ISO 11607 (Parts 1 and 2) is the internationally recognized standard for sterile barrier systems:
- Part 1: Covers material and system requirements
- Part 2: Covers validation of forming, sealing, and assembly processes
FDA-registered manufacturers align their validation documentation with this standard — and customs authorities frequently request it for international shipments.
GMP Compliance in Packaging Operations
Good Manufacturing Practice (GMP) regulations require that packaging materials are sourced, stored, and handled in controlled environments to prevent contamination. This applies not only to the packaging manufacturer but also to any 3PL or distribution partner handling packaged medical products.
Labeling and Traceability Requirements
Beyond storage and handling, FDA regulations specify what must appear on the packaging itself:
- Device name and manufacturer
- Sterility statement
- Expiration or use-by date
- Lot/batch number
- Special storage or handling instructions
Non-compliant labeling can result in customs holds or regulatory action.

International Shipping Compliance
Medical products shipped internationally must meet both the exporting country's regulations and the importing country's requirements. Key compliance requirements for cross-border shipments include:
- CE marking for EU market entry
- Packaging validation documents (frequently requested at customs)
- Country-specific labeling requirements for the importing market
A logistics partner with documented compliance capabilities can prevent costly customs delays — and protect product integrity throughout the journey.
Bluebonnet Medical Supplies operates with FDA-cleared handling, ISO compliance, and GMP-compliant processes, so medical product businesses can trust their inventory is stored, picked, and shipped without running into regulatory roadblocks.
How Packaging Choices Impact Medical Product Distribution
Shipping Costs and Carrier Rules
Packaging dimensions, weight, and fragility directly influence shipping rate categories. FedEx and UPS now round up every fractional inch when measuring packages, and FedEx applies an Additional Handling Surcharge for packages over 10,368 cubic inches. Flexible formats typically reduce DIM weight compared to rigid secondary cartons — a meaningful cost difference at volume.
Packaging must also withstand distribution conditions: drops, vibration, and stacking pressure. Products that arrive damaged trigger claims, returns, and reputational risk with buyers.
Sterile Barrier Integrity in Transit
Packaging integrity must be preserved not just at the point of manufacture but through warehousing (temperature, humidity controls), pick-and-pack handling, and last-mile delivery. A single compromised seal invalidates sterility and can expose a business to liability. A study of FDA Class I recalls found that packaging was the root cause in 47.4% of cases, often due to compromised sterile barriers.

Returns and Repackaging
When a seal fails in transit, the downstream problem is often a return. Returned medical products require careful inspection to determine whether packaging integrity has been maintained:
- Items with intact packaging may be eligible for restock after assessment
- Compromised packaging typically requires repackaging or disposal
- Documented inspection records protect against liability disputes
Bluebonnet's returns processing service assesses and restores eligible products rather than writing off inventory — recovering value that would otherwise be lost.
Customs Documentation
Medical products in flexible packaging require accurate commercial invoices, HS codes for medical devices, and sometimes a Certificate of Conformity or sterilization documentation to clear customs. Missing paperwork is a leading cause of customs delays for medical product shipments.
Frequently Asked Questions
What types of packaging are used for medical products?
Common formats include pouches (breathable and film-to-film), blister packs, IV bags, FFS films, and thermoformed trays. Each is selected based on the product type, required sterilization method (ETO, gamma, steam), and distribution environment.
What are the FDA requirements for pharmaceutical packaging?
Pharmaceutical packaging in the US must comply with 21 CFR regulations and, for sterile barrier systems, ISO 11607. Required labeling elements include the NDC number, lot number, expiry date, and storage conditions. Documented validation testing is required before market release.
What is the difference between flexible and rigid medical packaging?
Flexible packaging uses pliable films and laminates that conform to product shape, are lighter, and often lower cost to ship. Rigid packaging (hard plastic cases, glass vials) offers more structural protection but adds bulk and weight. The choice depends on the product's fragility, sterilization method, and distribution requirements.
What sterilization methods are compatible with flexible medical packaging?
Compatibility depends on the method:
- ETO: Requires breathable materials like Tyvek® or medical paper
- Gamma radiation: Compatible with most polymer films, including PE
- Steam/autoclave: Requires heat-tolerant materials like polypropylene
Each format must be validated for the specific sterilization cycle used.
How does packaging affect the shelf life of medical products?
A material's barrier properties — moisture, oxygen, and light resistance — directly determine how long a product stays sterile and effective. High-barrier laminates with aluminum foil extend shelf life significantly for moisture-sensitive or drug-coated devices compared to single-layer film.
Do medical packaging requirements differ for international shipping?
Yes, every importing country has its own framework. The EU requires CE marking and EU MDR compliance; other markets may require country-specific labeling or translated documentation. Customs agencies frequently request packaging validation records and certificates of conformity, so having these ready before shipment avoids clearance delays.


