
Introduction
Packaging single-use medical devices isn't just about putting a product in a bag. It means maintaining sterility standards, meeting FDA labeling requirements under 21 CFR Part 801, ensuring ISO 11607 compliance for sterile barrier systems, and managing supply chain handling—all at once.
A packaging failure during transit is a regulatory violation and a patient safety issue, not just a logistics problem. It can trigger FDA enforcement action.
This guide covers what single-use medical device packaging is, the types and materials involved, regulatory requirements, key benefits, sustainability considerations, and logistics best practices. It's designed for medical device manufacturers, online medical supply retailers, Amazon sellers of medical products, and businesses shipping medical devices domestically or internationally.
TLDR:
- Single-use devices require validated sterile barrier systems meeting ISO 11607 standards
- FDA considers devices without reprocessing instructions single-use by default—creating liability for third-party reprocessors
- Packaging must survive distribution stresses per ASTM D4169 testing protocols
- Studies report reprocessed single-use devices cause significant contamination rates and hundreds of documented infections
- FDA-cleared, ISO/GMP-compliant 3PLs prevent customs delays and packaging integrity failures
What Is Single-Use Medical Device Packaging?
Defining Single-Use Devices and Packaging Distinctions
The FDA defines a single-use device (SUD) as one "intended for use on one patient during a single procedure" and "not intended to be reprocessed (cleaned, disinfected/sterilized) and used on another patient." If a device's labeling lacks explicit reprocessing instructions—regardless of marketing claims—the FDA classifies it as single-use by default.
That default classification carries real liability. When hospitals or third parties reprocess SUDs, they legally become the manufacturer and must comply with all FDA labeling, premarket clearance, and quality system requirements.
The reprocessor must replace the original manufacturer's name and address on the label with their own — otherwise the device is considered "misbranded" under the Federal Food, Drug, and Cosmetic Act.
The Sterile Barrier System: Foundation of Single-Use Packaging
ISO 11139:2018 defines a "sterile barrier system" as the minimum packaging required to minimize microbial ingress risk and allow aseptic presentation at the point of use. As a validated system, it must maintain a Sterility Assurance Level (SAL) of 10⁻⁶ — less than one in a million chance of contamination — from manufacture through storage, shipping, and clinical use.
Single-Barrier vs. Dual-Barrier Sterile Packaging
Both approaches must meet ISO 11607 requirements, but they serve different distribution scenarios:
- Dual-barrier (two sealed layers, such as a pouch within a pouch): suited for heavy devices, international shipments, and high-risk distribution environments
- Single-barrier (one robust, validated layer): suited for lightweight devices, controlled distribution, and cost-sensitive applications where distribution testing confirms adequate protection
Both approaches must pass identical validation requirements, but single-barrier reduces material waste and packaging costs when distribution testing confirms sufficient protection.
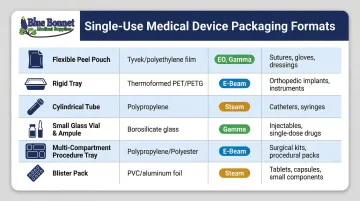
Common Types and Applications of Single-Use Medical Device Packaging
Not all single-use devices ship in the same format. The packaging type depends on device geometry, sterilization method, and end-use handling requirements. Here's how the main formats break down.
Flexible Pouches and Peel Pouches
The workhorse of high-volume consumables. One side uses clear film for visual identification; the other uses breathable Tyvek to allow EO or gamma sterilization. The chevron-style end seal supports aseptic opening at the point of use.
Common applications:
- Syringes and needles
- Surgical instruments
- Catheters and tubing
- Adhesive dressings and small surgical gloves
Rigid Trays
Thermoformed plastic trays — typically PETG or high-impact polystyrene — provide structural support for heavy or irregularly shaped devices. The device sits in a shaped cavity, sealed under a die-cut Tyvek lid to maintain sterility through distribution.
Common applications:
- Orthopedic implants
- Custom surgical procedure trays
- Multi-component device kits
- Dental and ophthalmic surgical tools
Sterile Tubes and Cylindrical Rigid Packaging
Long, narrow devices that don't fit standard trays require specialized cylindrical packaging. These rigid tubes feature custom end caps and internal cushioning to prevent movement during shipping.
Common applications:
- Intramedullary nails
- Spinal fixation components
- Endoscopic instruments (320mm–440mm lengths)
- Laparoscopic tools with delicate optics
Vials and Ampules with Tamper-Evident Seals
Sealed glass or plastic containers maintain chemical and biological stability for injectable medications and biologics. Under 21 CFR 211.132, tamper-evident packaging must provide visible evidence to consumers that tampering has occurred.
Common applications:
- Injectable medications
- Biologics and vaccines
- Liquid diagnostic reagents
- Sterile saline solutions
Custom Procedure Trays and Kit Packaging
Pre-assembled packages containing multiple sterile items for specific procedures. Each component within the kit must maintain individual sterility while the entire kit is packaged together for convenience.
Common applications:
- IV start kits
- Wound care kits
- Central line insertion trays
- Surgical prep kits
Blister Packs and Unit-Dose Packaging
Distinct from rigid trays, blister packs are designed for individual-dose protection — each unit sealed separately to maintain shelf life and prevent cross-contamination. The format is common in drug delivery systems where precise, tamper-evident unit isolation matters.
Common applications:
- Oral medications
- Inhalers
- Insulin pens
- Transdermal patches
Here's a quick reference across all six formats:
| Format | Primary Material | Sterilization Compatible | Best For |
|---|---|---|---|
| Flexible/Peel Pouches | Clear film + Tyvek | EO, gamma | High-volume consumables |
| Rigid Trays | PETG, HIPS + Tyvek lid | EO, gamma | Heavy or shaped devices |
| Cylindrical Tubes | Rigid plastic + end caps | EO, gamma | Long, narrow instruments |
| Vials and Ampules | Glass or plastic | Terminal sterilization | Injectables, biologics |
| Custom Procedure Trays | Mixed materials | EO, gamma | Multi-component kits |
| Blister Packs | Formed plastic + foil/film | EO, radiation | Unit-dose medications |
Regulatory Requirements and Labeling Compliance
FDA Labeling Requirements Under 21 CFR Part 801
All single-use medical device packaging sold in the US must comply with FDA labeling requirements. Non-compliant packaging is "misbranded" and subject to enforcement action.
Mandatory label elements:
- Name and address of manufacturer, packer, or distributor
- Common or usual name of the device
- Net quantity of contents
- Adequate directions for use (or applicable exemption)
- Adequate warnings and precautions
- No false or misleading statements
21 CFR 801.20 also requires a Unique Device Identifier (UDI) on every device label and package, presented in both plain-text and Automatic Identification and Data Capture (AIDC) format such as a barcode.
ISO 11607: International Standard for Sterile Packaging
ISO 11607 governs sterile packaging for terminally sterilized medical devices worldwide. Most international markets—including the US and EU—recognize it as the baseline standard for sterile barrier systems. The standard has two parts:
- ISO 11607-1:2019: Covers materials, sterile barrier systems, and packaging systems—specifically their ability to maintain sterility until the point of use.
- ISO 11607-2:2019: Covers validation of forming, sealing, and assembly processes used during packaging production.

Manufacturers must validate that packaging withstands distribution stresses (vibration, compression, temperature changes) while maintaining sterile barrier performance and material compatibility.
Reprocessing and Labeling Responsibility
If a hospital or third-party facility reprocesses a single-use device, they legally become the manufacturer. The reprocessor must:
- State their own name and place of business on the label (not copy the original manufacturer's label)
- Meet all FDA premarket clearance requirements
- Implement a complete quality system
- Assume all liability for device performance and safety
Original manufacturers are shielded from liability once a device leaves their control—but only if their own labeling and documentation are correctly maintained from the start.
Sterilization Method and Packaging Material Alignment
The sterilization method dictates packaging material selection:
EtO (Ethylene Oxide) Gas Sterilization:
- Requires breathable packaging (Tyvek, medical-grade paper)
- Gas must penetrate, sterilize, and outgas to safe residual levels
- Tyvek outgases faster than medical-grade paper, reducing toxic residuals
Gamma Radiation:
- Compatible with most materials but may degrade polymers
- Requires maximum acceptable dose (D max,acc) testing per ISO 11137-1:2015
- Cumulative radiation effects can cause embrittlement or discoloration
Steam Autoclave:
- Requires heat-tolerant, moisture-permeable materials
- Tyvek withstands controlled steam at 121°C–127°C
- Standard plastics may melt or warp
International Regulatory Requirements
If you ship devices beyond the US, destination-country requirements add another layer of documentation. The EU MDR 2017/745 is the most comprehensive framework outside the US—and non-compliance can halt customs clearance entirely.
EU MDR 2017/745 Requirements:
For devices exported to the European Union, sterile packaging must display:
- Indication permitting recognition as sterile packaging
- Declaration that device is in sterile condition
- Method of sterilization used
- Month and year of manufacture
- Unambiguous time limit for safe use
- Instructions if sterile packaging is damaged
- CE marking affixed to device or sterile packaging
For businesses shipping internationally, getting packaging documentation right before products leave the warehouse prevents costly delays at customs. Bluebonnet Medical Supplies handles FDA-compliant packaging, GMP/ISO-aligned storage, and international shipment documentation for medical product companies that need their logistics managed correctly from the start.
Key Benefits of Single-Use Medical Device Packaging
Sterility and Patient Safety
Individually sealed single-use packaging eliminates cross-contamination risk between patients and removes the need for sterilization verification at point of care. This directly reduces healthcare-associated infections (HAIs).
The contamination risk with reprocessed devices is well-documented:
- Research shows a 15.25% contamination rate in reprocessed patient-ready duodenoscopes
- Outbreaks linked to contaminated duodenoscopes have caused 490 infections and 32 deaths

Single-use packaging with validated sterile barriers removes this failure point from the equation — no reprocessing cycle means no reprocessing failure.
Operational Efficiency
Single-use packaging eliminates labor-intensive reprocessing steps—cleaning, sterilizing, re-inspecting, and tracking. For medical product businesses, this means:
- Fewer returns due to contamination concerns
- Reduced complaint handling
- Lower liability exposure
- Faster time-to-use in clinical settings
Total Cost of Ownership Advantages
Activity-based costing analysis shows reusable equipment costs $1,019 per case versus $917 for single-use alternatives — meaning single-use is often cheaper overall, despite the higher per-unit price.
Hidden costs of reusable devices include:
- Capital purchase and depreciation
- Maintenance, repair, and backup systems
- Reprocessing labor and time
- Sterilization supplies and facility costs
- Operating room setup time
For medical product businesses, understanding this total cost picture matters — customers who grasp the real economics of single-use are easier to retain and less likely to revert to reusable alternatives.
Materials Used and Sustainability Considerations
Common Packaging Materials
Material selection must balance sterilization compatibility, barrier properties, and regulatory compliance:
- Polyethylene (PE) and Polypropylene (PP) — flexible film for pouches and wraps
- Tyvek (HDPE nonwoven) — breathable sterile barrier for EtO sterilization
- Polyvinyl Chloride (PVC) — flexible packaging with good clarity
- PETG and High-Impact Polystyrene (HIPS) — thermoformed rigid trays
- Medical-grade paper — breathable barrier for select applications
The Sustainability Challenge
Medical packaging regulations prioritize patient safety over environmental considerations. The healthcare sector generates over 32 billion pounds of plastic waste annually, projected to reach 48 billion pounds by 2025. The vast majority of this uncontaminated, recyclable material is currently landfilled or incinerated.
The industry is exploring bioplastics (starch-based, cellulose, chitosan) as alternatives, but scalability and sterility validation remain barriers. For example, exposing chitosan to 50 kGy of gamma irradiation decreases its molecular weight by 79.24%, destroying its structural integrity. No current bioplastic survives terminal sterilization without severe degradation — leaving them non-compliant under ISO 11607 standards.
Practical Sustainability Steps
Until viable alternatives emerge, medical product businesses can still reduce environmental impact through practical design choices:
- Right-size packaging to cut excess material from the start
- Use recyclable materials where sterility requirements permit
- Specify recycled or biodegradable materials for outer, non-sterile protective packaging
- Design for mechanical recycling following Healthcare Plastics Recycling Council (HPRC) guidelines
Storage, Handling, and Shipping of Single-Use Medical Devices
Critical Storage Conditions
Even properly validated packaging can be compromised by improper storage. Required conditions for most sterile single-use devices include:
- Controlled temperature ranges (typically 68°F–72°F)
- Low humidity (30%–50% relative humidity)
- Protection from direct sunlight and UV exposure
- Protection from physical damage (crushing, puncture)
- Clean, organized storage preventing contamination
ISO 11607-1 requires manufacturers to demonstrate that material properties remain within validated limits during storage under specified conditions.
Distribution Stress Testing Requirements
Packaging must be tested for distribution stresses—vibration, compression, and environmental changes during shipping. Industry standards include:
ASTM D4169 — FDA-recognized standard providing uniform basis for evaluating shipping units through laboratory simulation of anticipated hazard elements (drops, compression, vibration) in various distribution cycles.
ISTA Protocols:
- ISTA 2A: Partial simulation for packaged products weighing 150 lbs or less
- ISTA 3A: Advanced general simulation for parcel delivery shipments
- ISTA 7E: Thermal transport packaging testing for external temperature exposures

Together, these protocols confirm that packaging holds its sterile barrier integrity from warehouse to final destination.
Compliant Medical Device Logistics in Practice
Proper medical device logistics requires:
- Temperature-controlled warehousing with environmental monitoring
- FIFO inventory management to ensure proper product rotation
- Accurate documentation for customs and regulatory purposes
- Appropriate carrier selection for sensitive medical goods
- Compliance expertise to navigate international requirements
Meeting all of these requirements is where a specialized 3PL partner makes a tangible difference. Bluebonnet Medical Supplies handles storage, packaging, and shipping under FDA, ISO, and GMP compliance standards — with carrier relationships that cut costs and customs experience that keeps shipments moving.
Frequently Asked Questions
Can single-use medical devices be reprocessed and reused?
FDA regulations allow reprocessing of some SUDs, but the reprocessor becomes the legal manufacturer and must meet all labeling, premarket clearance, and quality system requirements. Cleaning and repackaging alone is not sufficient. The reprocessor assumes full regulatory burden and liability.
What labeling is required on single-use medical device packaging?
Mandatory elements per 21 CFR Part 801 include: manufacturer name and address, device name, quantity of contents, directions for use (or applicable exemption), adequate warnings, and a UDI in both plain-text and barcode format.
What is the difference between single-barrier and dual-barrier sterile packaging?
Dual-barrier uses two sealed layers for added protection against physical damage, while single-barrier uses one robust, validated layer. Both must meet ISO 11607 standards, but single-barrier reduces material waste and cost when distribution testing confirms adequate protection.
How should single-use medical devices be disposed of after use?
Most single-use device packaging can be discarded as regular waste. However, items contaminated with blood or bodily fluids, or sharps, are regulated medical waste requiring special handling per federal, state, and local regulations.
What sterilization methods are compatible with single-use medical device packaging?
Three methods are widely used:
- EtO gas — requires breathable material like Tyvek
- Gamma radiation — compatible with most materials, though it may degrade some polymers
- Steam autoclave — requires heat-tolerant materials
Both the device and packaging must be validated as compatible with whichever method is chosen.
How do you ensure packaging integrity is maintained during shipping?
Packaging must be distribution-tested per ASTM D4169 or ISTA protocols to validate survival of shipping stresses. Proper warehousing conditions, appropriate carrier selection, and partnering with a compliant medical logistics provider all play critical roles in protecting sterile integrity during transport. Bluebonnet Medical Supplies offers FDA-compliant fulfillment and shipping services built for exactly these requirements.


