
Introduction
A single mislabeled product can trigger an FDA warning letter, a costly recall, or irreversible damage to consumer trust. FDA food labeling requirements exist to protect consumers from allergens, misleading claims, and unsafe products. They also create a consistent, enforceable standard for every business selling in the U.S. market.
This guide covers the core components of FDA labeling compliance:
- What the FDA considers "labeling" under 21 CFR
- Mandatory label elements and Nutrition Facts panel rules
- Category-specific requirements and permitted claims
- The most common mistakes that trigger enforcement action
It's written for food manufacturers, distributors, and businesses handling regulated products who need a clear picture of compliance requirements — whether approaching this for the first time or auditing existing labels.
TLDR
- "Labeling" includes websites and brochures—not just the physical package
- Nine major allergens must be declared (including sesame as of 2023)
- Nutrition Facts panels must list Added Sugars and include updated Daily Value footnotes
- Dual-column labels are mandatory for packages containing 200-300% of serving size
- Small business exemptions vanish if you make any nutrient or health claim
- FDA does not pre-approve food labels—manufacturers self-certify compliance
What Does the FDA Consider "Labeling"?
The FDA draws a legal distinction between a "label" and "labeling." A label is the physical tag or wrapper affixed directly to a product container. Labeling is far broader.
Under the Federal Food, Drug, and Cosmetic Act (FD&C Act), labeling includes all written, printed, or graphic materials accompanying a product in commerce—brochures, product inserts, and even online grocery product pages.
Governing Regulations
Food labeling is primarily governed by:
- Title 21 of the Code of Federal Regulations (CFR), Part 101 – General food labeling standards
- Nutrition Labeling and Education Act (NLEA) – Mandates Nutrition Facts panels
- Food Safety Modernization Act (FSMA) – Traceability and preventive controls
In May 2025, the FDA updated Compliance Program 7321.005, expanding its scope to both domestic and import food labeling inspections. Inspectors now prioritize undeclared allergens, missing nutrition labeling, and non-compliant claims.
Key Takeaway: Your e-commerce product pages are legally considered labeling. All mandatory information—allergens, ingredients, and Nutrition Facts—must be accessible to consumers before they complete a purchase.
Mandatory Information on FDA Food Labels
Principal Display Panel (PDP)
The PDP is the part of the label most likely to be seen at the point of purchase. It must prominently display:
- Statement of Identity: The common or usual name of the product (e.g., "Orange Juice," not "Morning Refresher"), displayed in bold type proportional to the most prominent printed matter
- Net Quantity of Contents: Weight, volume, or count (e.g., "12 oz," "355 mL," "24 cookies"), placed in the bottom 30% of the PDP with minimum type size based on PDP area (e.g., 1/8 inch for 5–25 sq. inches)
Information Panel
The PDP captures attention, but the Information Panel—immediately to the right of the PDP as the consumer sees it—carries the detailed disclosures. It must include:
- Manufacturer/Distributor Information: Name and complete address (street, city, state, ZIP code), plus country of origin if imported
- Ingredient List: All ingredients listed from highest to lowest amount by weight, with sub-ingredients in parentheses (e.g., "Enriched Flour (Wheat Flour, Niacin, Iron)"). Minimum type size is 1/16 inch (measured by the height of a lowercase letter). Spices, flavors, and colors may be declared collectively (e.g., "natural flavor"), but any allergen within them must still be declared explicitly.
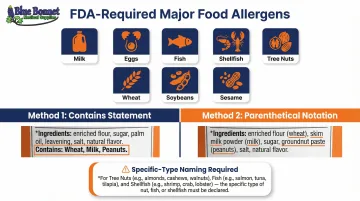
Allergen Declaration
Under FALCPA and the FASTER Act, nine major allergens must be declared: milk, eggs, fish, shellfish, tree nuts, peanuts, wheat, soybeans, and sesame. Manufacturers can choose one of two disclosure methods:
- "Contains" statement: Immediately after the ingredient list (e.g., "Contains: Milk, Wheat, Sesame")
- Parenthetical notation: Within the ingredient list (e.g., "Whey (Milk)")
For tree nuts, fish, and shellfish, the specific type must be named (e.g., almonds, flounder, crab).
Nutrition Facts Panel is required for most packaged foods (covered in full in the next section).
Expiration Dating, Lot Numbers, and Traceability
Dating and traceability rules are more nuanced than most manufacturers expect:
- "Use-by" dates are mandatory only for infant formula under 21 CFR Part 107
- Quality dating ("Best if Used By") is voluntary for other foods but FDA-recommended to reduce waste
- Lot/batch codes are mandatory for acidified and low-acid foods under 21 CFR 114.80
- FSMA Traceability Lot Codes (TLCs) are required for certain high-risk foods under 21 CFR Part 1 Subpart S (enforcement begins July 2028); TLCs do not appear on the consumer label — they belong in supply chain records only
The Nutrition Facts Panel: Requirements and Format
The 2016 Nutrition Facts final rule overhauled how nutrition information must appear on food packaging — from serving size calculations to which nutrients are required. Understanding the current format requirements is the first step toward full FDA compliance.
Standard Format Requirements
Serving Size
- Must reflect Reference Amounts Customarily Consumed (RACCs) based on what people actually eat, not manufacturer preference
- RACCs are listed in 21 CFR 101.12
Calories
- Displayed in large, bold type
- "Calories from Fat" has been removed
Mandatory Nutrients (in order):
- Total Fat, Saturated Fat, Trans Fat
- Cholesterol
- Sodium
- Total Carbohydrates, Dietary Fiber, Total Sugars, Added Sugars (indented under Total Sugars)
- Protein
- Vitamin D, Calcium, Iron, Potassium (with quantitative amounts and %DV)
Daily Value (DV) Footnote Must state exactly: "The % Daily Value tells you how much a nutrient in a serving of food contributes to a daily diet. 2,000 calories a day is used for general nutrition advice."
Dual-Column Labeling Rules
Packages containing 200-300% of the RACC must display both per-serving and per-container nutrition information side by side. This applies to:
- Products typically consumed in one sitting (for example, a 20 oz soda bottle)
- Individual units weighing 200-300% of RACC (for example, a large muffin)
| Package Size vs. RACC | Label Format Required |
|---|---|
| < 200% of RACC | Single-serving container (entire content = one serving) |
| 200-300% of RACC | Dual-column required |
| > 300% of RACC | Single column showing per-serving info |
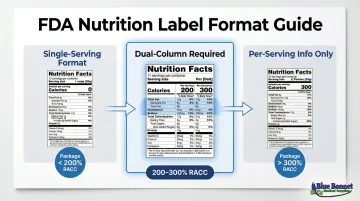
Exception: Small packages using tabular or linear format due to limited space (<40 sq. inches) are exempt from dual-column requirements.
Small Business Exemptions
Under 21 CFR 101.9(j), small businesses may qualify for exemptions from Nutrition Facts labeling:
| Exemption Type | Threshold Criteria | Filing Requirement |
|---|---|---|
| Retailer Exemption | Annual gross sales ≤ $500,000 OR food sales ≤ $50,000 | No FDA notice required |
| Low-Volume Product | < 100 FTE employees AND < 100,000 units sold annually | Annual notice must be filed with FDA |
| Micro-Business | < 10 FTE employees AND < 10,000 units sold annually | No FDA notice required |
One exception overrides all of these: Making any nutrient content claim (such as "sugar free"), any health claim, or displaying nutrition information anywhere on the label or in advertising immediately voids the exemption — at which point full Nutrition Facts labeling is required.
FDA Food Labeling Requirements by Product Category
Standard Packaged Foods
Most packaged foods must include all mandatory elements described above. However:
- Restaurant-style or bulk foods sold without packaging have lighter requirements
- Online food retailers must ensure all mandatory labeling information is accessible to consumers before purchase
Dietary Supplements
Dietary supplements are regulated under the Dietary Supplement Health and Education Act (DSHEA). They require:
- Supplement Facts panel (not Nutrition Facts)
- List of dietary ingredients and amounts per serving
- Serving size
- Mandatory disclaimer: "This statement has not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure, or prevent any disease."
- No disease claims permitted
Labels must remain intact and legible through the entire distribution chain — from warehouse to end consumer. This is especially relevant for supplement brands managing third-party fulfillment or cross-state shipping.
Infant Formula and Foods for Special Dietary Use
Infant formula is subject to the strictest food labeling rules under 21 CFR Part 107, requiring:
- Specific nutrient declarations within defined ranges
- Preparation instructions with pictograms
- Mandatory "Use-by" date guaranteeing nutrient quantity and quality until that date
Medical foods and foods for special dietary use (such as hypoallergenic formulas) have additional labeling rules and are exempt from certain Nutrition Facts requirements.
Approved Health and Nutrient Content Claims
The FDA strictly regulates front-of-pack claims. Making an unauthorized claim renders a product misbranded.
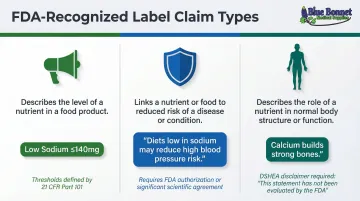
Three FDA-Recognized Claim Types
1. Nutrient Content Claims
- Characterize the level of a nutrient (such as "low sodium," "high fiber," or "sugar free")
- Must meet specific quantitative thresholds defined in 21 CFR Part 101 Subpart D
- Examples: "Low sodium" = ≤140 mg per serving; "High fiber" = ≥5 g per serving
2. Health Claims
- Describe a relationship between a nutrient or food and reduced risk of disease
- The FDA must authorize these based on significant scientific agreement, or the claim must qualify with a disclaimer
- Example: "Diets low in sodium may reduce the risk of high blood pressure"
3. Structure/Function Claims
- Describe the role of a nutrient in normal body function (for example, "calcium builds strong bones" or "fiber maintains bowel regularity")
- These don't require FDA pre-approval, but claims must be truthful and stop short of implying disease prevention. Supplement makers must also include the DSHEA disclaimer and submit a 30-day FDA notice.
Understanding what each claim type allows is only half the equation — knowing what triggers enforcement is equally important.
What Makes a Claim "Misleading"
The FDA targets several patterns when pursuing misbranding enforcement:
- Images of hearts or similar symbols implying cardiovascular benefits without an authorized claim
- Manipulated serving sizes designed to make nutrient numbers look more favorable
- Front-of-pack language that contradicts the Nutrition Facts panel — for example, "low sugar" on a product with 15g per serving
Two specific claims — "healthy" and "natural" — deserve extra attention because their regulatory status is not what most manufacturers expect.
"Healthy" and "Natural" Claims
"Healthy" (Finalized December 2024): The FDA finalized an updated definition of the "healthy" nutrient content claim effective February 2025 (compliance date February 2028). The new framework requires:
- Foods must contain Food Group Equivalents (FGEs) from vegetables, fruits, whole grains, dairy, or protein
- Defined caps on added sugars, saturated fat, and sodium based on %DV
"Natural" (Undefined): The FDA has no formal regulatory definition for "natural." The agency's informal position holds that "natural" means nothing artificial or synthetic has been added — but because this is never codified, the term carries significant litigation risk for manufacturers who lean on it.
FDA Pre-Approval Misconception
The FDA does not pre-approve most food labels. Manufacturers are responsible for self-certifying compliance with all applicable regulations before going to market. The FDA reviews labels during inspections or in response to complaints.
Common FDA Food Labeling Mistakes (and How to Avoid Them)
Frequent Violations Triggering Warning Letters
1. Missing or Incorrect Allergen Declarations
Undeclared allergens are the leading cause of FDA recalls. Recent enforcement actions highlight this risk:
- Stew Leonard's (November 2024): Fatal anaphylactic reaction linked to cookies that failed to declare peanuts and eggs. Warning letter issued.
- Bimbo Bakeries (June 2024): Listed allergens (sesame, walnuts, almonds) in the "Contains" statement that were not in the product formulation—using precautionary labeling defensively instead of proper allergen controls.

How to Avoid: Implement allergen preventive controls under 21 CFR Part 117. Verify vendor-printed labels against current formulas before use.
2. Failure to List Added Sugars Separately
The 2016 rule mandates that Added Sugars be indented under Total Sugars with a %DV. Many products still omit this.
3. Illegible Font Sizes
FDA mandates minimum type sizes based on label surface area. The Information Panel requires at least 1/16-inch type height; net quantity type size varies by PDP area.
4. Missing Net Quantity or Manufacturer Information
Net quantity must appear in the bottom 30% of the PDP. Manufacturer name and address must be complete (street, city, state, ZIP).
Claims-Related Errors
Using Undefined Terms:
- "Natural," "light," "healthy" without meeting FDA's specific definitions (or using terms that have no definition)
- Making unapproved health claims (e.g., "prevents heart disease")
- Listing structure/function claims without the required DSHEA disclaimer (for supplements)
Misleading Front-of-Pack Language: The FDA increasingly scrutinizes implied claims. For example, images of fruit on a product with no actual fruit content, or "made with whole grains" when whole grains are a minor ingredient.
Multilingual and International Labeling Pitfalls
If a label contains any representation in a foreign language, all required statements must appear in both English and the foreign language. Inaccurate translations of allergen warnings or ingredient names are a critical risk area.
Products exported to other countries must meet both FDA requirements and the destination country's labeling laws. Engaging a logistics partner experienced in regulated product shipping—and consulting a regulatory attorney familiar with destination-country requirements—can help prevent costly customs delays.
Frequently Asked Questions
What information is mandatory on FDA food labels?
Six core elements are required under 21 CFR Part 101: statement of identity (product name), net quantity of contents, manufacturer/distributor name and address, ingredient list in descending order by weight, Nutrition Facts panel, and allergen declarations for the nine major allergens.
What are the FDA packaging and labeling requirements?
FDA packaging requirements (21 CFR Parts 174-186) ensure materials are safe for food contact. Labeling requirements dictate what information must appear on the package. These are distinct compliance obligations—packaging safety and label content are both mandatory.
What does the FDA consider "labeling"?
Under the FD&C Act, "labeling" covers all written, printed, or graphic materials accompanying a product in commerce, including brochures, promotional materials, and online product pages directly linked to the product at the point of sale.
What is FDA-approved labeling?
Unlike drugs or medical devices, most food labels do not require FDA pre-approval. Manufacturers are responsible for self-certifying compliance with 21 CFR requirements. The FDA may review and act on labels during inspections or in response to consumer complaints.
What are the FDA labeling requirements for dietary supplements?
Dietary supplements require a Supplement Facts panel (not Nutrition Facts), a full ingredient list, serving size, and the DSHEA disclaimer stating the product is not intended to diagnose, treat, cure, or prevent disease. Drug or disease claims are prohibited under DSHEA and 21 CFR Part 101.
What should diabetics look for on food labels?
Diabetics should focus on total carbohydrates, dietary fiber, added sugars, and serving size on the Nutrition Facts panel. Also scan the ingredient list for hidden sugar sources such as corn syrup, dextrose, and maltose — all required disclosures on FDA-compliant labels.
Final Takeaway: FDA food labeling compliance is a manufacturer responsibility, not an FDA pre-approval process. Stay current with 21 CFR Part 101, monitor FDA warning letters for emerging enforcement priorities, and ensure every element—from allergen declarations to Nutrition Facts formatting—is accurate before your product reaches consumers.


