
Introduction
Wellness products—supplements, vitamins, probiotics, and personal care items—occupy a regulated middle ground between standard consumer goods and pharmaceuticals. Shipping them in the US involves far more complexity than typical e-commerce: product integrity can degrade in hours, FDA oversight is real and enforced, and consumer safety is non-negotiable.
The risks of getting compliance or storage wrong are severe. Product degradation leads to costly recalls, FDA violations trigger warning letters and product seizures, and lost customer trust can permanently derail a growing brand. The average direct cost of a consumer product recall is approximately $10 million, with indirect costs—brand damage, lost contracts, litigation—often multiplying that figure.
Avoiding that outcome starts with understanding the rules. What follows breaks down the FDA and DSHEA requirements that govern wellness products, the storage conditions that protect quality, the shipping practices that maintain compliance from warehouse to doorstep, and the mistakes that catch even experienced brands off guard.
TL;DR
- Wellness products sold in the US fall under the FDA's DSHEA framework and must meet GMP standards throughout storage and fulfillment—not just manufacturing
- Storage conditions directly affect product quality, expiration compliance, and recall readiness—temperature and humidity failures cause potency loss and texture degradation
- Labeling, lot tracking, and documentation are legal requirements with real liability consequences if skipped
- Temperature-sensitive products like probiotics require cold-chain handling; choosing the wrong carrier or packaging compromises efficacy
- Partnering with a GMP-compliant 3PL reduces compliance risk while cutting shipping costs through established carrier relationships
Why Shipping Compliance Matters for Wellness Products
Wellness products are subject to FDA oversight under the Dietary Supplement Health and Education Act (DSHEA), which creates real legal obligations that many DTC brands and Amazon sellers underestimate. Unlike standard consumer goods, supplements must meet Current Good Manufacturing Practice (CGMP) standards at every stage—manufacturing, storage, packaging, and fulfillment.
The FDA actively enforces compliance through warning letters and product seizures. In 2021 and 2023, U.S. Marshals seized adulterated dietary supplements in multi-state operations. Industry data shows that the average direct cost for a food or supplement recall is $10 million, excluding litigation and brand damage—which can erase over $100 million in shareholder value within days.
Those financial risks often trace back to warehouse conditions. Poor storage or handling upstream directly causes downstream compliance failures, and the damage is usually invisible until the customer opens the package. Common examples include:
- Probiotics stored at improper temperatures that lose CFU potency but still ship
- Gummies exposed to heat that melt and clump in transit
- Powders that absorb moisture and become unusable before they arrive
Consumers buying wellness products are particularly sensitive to quality claims. They're purchasing based on efficacy promises—probiotics that support gut health, vitamins that boost immunity, supplements that enhance performance. A single high-profile quality failure can permanently damage a brand's reputation in a category where repeat purchases depend entirely on delivered results.
Understanding FDA and Regulatory Requirements for Wellness Products
The Dietary Supplement Health and Education Act of 1994 (DSHEA) established the regulatory framework for wellness products in the US. Under DSHEA, the FDA does not approve dietary supplements for safety and effectiveness before they are marketed. Instead, manufacturers and distributors bear the initial responsibility for ensuring their products meet safety standards and are not adulterated or misbranded.
While DSHEA does not require pre-market approval, it mandates that firms follow Current Good Manufacturing Practices (CGMPs) to ensure the identity, purity, quality, strength, and composition of dietary supplements. These CGMP requirements extend beyond manufacturing — they apply equally to packaging, labeling, and holding (storage) of dietary supplements.
GMP Compliance in Storage and Fulfillment
21 CFR Part 111 outlines the CGMPs for dietary supplements. A common misconception is that 3PLs and storage facilities are exempt from these requirements. They are not.
Who Must Comply
21 CFR 111.1(b) explicitly states that the "retail exemption" does not apply to warehouses. A retail establishment does not include a warehouse or other storage facility for a retailer, or a warehouse that sells directly to individual consumers. Therefore, 3PLs that hold dietary supplements must comply with specific holding requirements.
Key GMP Requirements for Warehouses
GMP-compliant warehouse operations must include:
- Temperature, humidity, and light controls that protect product identity, purity, and strength
- Ventilation systems that minimize odors and vapors
- Written SOPs for all holding and distributing operations, including label-specific storage instructions
- Continuous environmental monitoring with documented temperature and humidity records
- Physically segregated storage areas to prevent cross-contamination between product types
- Personnel trained to handle regulated products under GMP standards

The FDA actively enforces these holding provisions. In a 2023 Warning Letter to Healthtex Distributors, Inc., the FDA cited the firm for failing to establish written procedures for holding and distributing operations, specifically noting a failure to include instructions on how to appropriately store products based on storage temperatures indicated on product labels.
Labeling Requirements for Wellness Products
Labeling errors are a primary driver of FDA enforcement actions. Dietary supplement labels must include specific mandatory elements:
- Statement of identity that clearly identifies the product as a dietary supplement
- Supplement Facts panel enclosed in a box with proper formatting
- Net quantity of contents (accurate weight or volume)
- Full ingredient list in descending order of predominance by weight
- Name and address of the manufacturer, packer, or distributor
The Required Disclaimer
If a product makes a structure/function claim (e.g., "calcium builds strong bones"), the entity must notify the FDA within 30 days of marketing and include a mandatory disclaimer. The exact, verbatim disclaimer text required by 21 CFR 101.93 is:
"This statement has not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure, or prevent any disease."
Failure to adhere to these formatting and content rules results in misbranding. For instance, Formulation Technology, Inc. received a 2024 Warning Letter because their "Supplement Facts" title was not set at full width, lacked required heavy/light bars, and featured incorrect serving sizes.
Lot Tracking, Expiration Management, and Recalls
Effective lot tracking is a foundational GMP requirement that enables targeted recalls and prevents expired product distribution.
Lot Number Requirements
Under 21 CFR 111.415(f), firms must assign a batch, lot, or control number to each lot of packaged and labeled dietary supplement distributed. Furthermore, 21 CFR 111.83 requires firms to collect and hold reserve samples of each lot distributed, identified with the batch/lot number, and retained for one year past the shelf life date or two years from the date of distribution.
FIFO vs. FEFO Inventory Rotation
While the FDA requires "appropriate conditions" and traceability, it does not explicitly mandate a specific inventory rotation method. However, industry best practices distinguish between two approaches:
| Method | Basis | Best For |
|---|---|---|
| FIFO (First-In-First-Out) | Ships oldest inventory by receipt date | General goods with uniform shelf life |
| FEFO (First-Expire-First-Out) | Ships inventory closest to expiration first | Wellness and dietary supplement products |
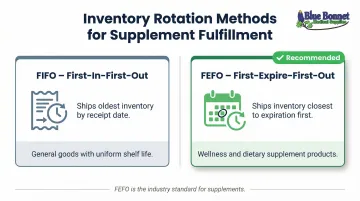
FEFO is the industry standard for wellness products because degradation ties to thermal history and elapsed time — not just when a unit arrived. Expiration-based rotation ensures products reach consumers well before efficacy degrades.
Recall Readiness
A compliant recall process requires the ability to trace all affected units by lot number, notify retailers or customers, and document the removal. Without proper lot tracking infrastructure, a recall becomes chaotic and costly. A 3PL that maintains real-time lot-level visibility — with documented chain of custody from receiving through shipment — is what makes the difference between a controlled withdrawal and a full-scale crisis.
Proper Storage Conditions for Wellness Products
Wellness products vary significantly in their storage needs—from standard dry storage for tablets to refrigerated conditions for probiotics. Failing to match storage conditions to product requirements is a direct GMP violation that compromises efficacy and triggers quality failures.
Temperature and Climate Control
The United States Pharmacopeia (USP) defines specific storage temperature categories that serve as industry standards:
| USP Storage Category | Temperature Range | Notes |
|---|---|---|
| Controlled Room Temperature (CRT) | 20° to 25°C (68° to 77°F) | Excursions allowed between 15° and 30°C (59° and 86°F). Mean Kinetic Temperature must not exceed 25°C. |
| Refrigerator / Controlled Cold | 2° to 8°C (36° to 46°F) | Required for probiotics and some functional beverages. |
| Freezer | -25° to -10°C (-13° to 14°F) | Temperature controlled to ±10°C. |
Temperature Excursion Risks
Temperature excursions have documented, measurable impacts on supplement quality:
- Probiotic viability drops significantly at higher temperatures; studies show that while strains may survive well at 5°C or 25°C, storage at 40°C results in detrimental CFU loss after just three months
- Gelatin-based softgels and gummies are highly susceptible to heat, with gelatin melting points ranging from 21–34°C depending on the formulation
- Potency loss in vitamins and active ingredients accelerates with each degree above recommended storage temperature
- Texture changes including clumping powders, melted gummies, and swollen capsules
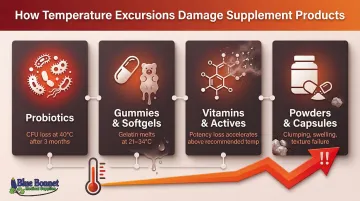
Even short exposures to improper temperatures can trigger quality failures that aren't immediately visible but become apparent when customers open the product.
Humidity, Ventilation, and Environmental Monitoring
Humidity control is just as critical as temperature. USP <659> defines a "Dry place" as a location that does not exceed 40% average relative humidity at 20°C (68°F).
What High Humidity Does to Wellness Products
Left unchecked, elevated humidity causes severe product failures:
- Caking and clumping — moisture-induced caking in carbohydrate-sensitive beverage powders increases with higher water activity
- Microbial growth — elevated relative humidity above 69% causes rapid visual deterioration and microbial proliferation in seed-based powders
- Softgel leakage — gelatin soft capsules absorb moisture in high humidity, leading to swelling, deterioration of mechanical properties, and eventual leakage
Environmental Monitoring Under FDA/GMP Rules
Under 21 CFR 111.455, the FDA requires holding components and supplements under "appropriate conditions of temperature, humidity, and light." The FDA does not mandate a single numeric RH target across all products, but storage conditions must be supported by stability data to prevent degradation.
GMP-compliant warehouses must maintain continuous environmental monitoring with documented logs available for audit. Many general-purpose warehouses fail wellness clients at exactly this point—they lack the monitoring infrastructure to demonstrate compliance when it matters.
Inventory Segregation and Storage Organization
GMP requires physical separation of different product types to prevent cross-contamination. This includes:
- Liquids separated from powders — preventing moisture contamination
- Supplements separated from non-supplement goods — avoiding cross-contact with non-food items
- Quarantine zones for returned or suspect inventory — physically isolating products under investigation until disposition is determined
Bluebonnet Medical Supplies warehouses wellness and medical products under these exact protocols—FDA-cleared packaging, GMP and ISO-compliant storage, and documented environmental monitoring built into daily operations. For brands that need compliant storage without building it in-house, that infrastructure is already in place.
Shipping Best Practices and Common Compliance Mistakes
Shipping compliance for wellness products extends beyond warehouse operations into carrier selection, packaging, and documentation.
Key Shipping Compliance Requirements
- Choose carriers with experience in wellness and supplement shipments who understand regulatory requirements
- Use tamper-evident seals, moisture-resistant materials, and insulated packaging for cold-chain items
- Ensure shipping documentation accurately reflects product contents and lot numbers
- Maintain cold-chain integrity with refrigerated or insulated packaging and temperature monitoring for sensitive products
Common Compliance Mistakes
Watch for these frequent errors that trigger FDA scrutiny or marketplace penalties:
- No cold-chain packaging for temperature-sensitive products — probiotics and functional beverages require insulated or refrigerated packaging to maintain potency
- Using a general-purpose 3PL without GMP credentials — standard fulfillment centers lack the environmental controls and documentation systems required for supplements
- Skipping FEFO controls on near-expiry inventory — sending soon-to-expire products violates both FDA expectations and marketplace policies
- Missing or incorrect labels — labeling errors are the most common reason wellness products face FDA warning letters

Documentation Requirements During Transit
Packing slips should include lot numbers and expiration dates for every shipment. Returns processes need a defined workflow for inspecting received inventory and categorizing units as sellable, quarantined, or disposable. That paper trail is what makes recall readiness and FDA compliance achievable.
Amazon FBA Requirements
For brands shipping through Amazon FBA or Seller Fulfilled Prime, Amazon has its own expiration date and labeling requirements that layer on top of FDA rules. Key requirements include:
- Products must have sufficient remaining shelf life at fulfillment center check-in to allow full consumption plus an additional 90 days
- Herbal and mineral supplements must arrive with at least 730 days before expiry
- Sellers must work with a third-party Testing, Inspection, and Certification (TIC) provider, with manufacturing verified by an accredited third-party audit for cGMP compliance
- Products not meeting these requirements face removal or disposal fees—standard-size removal runs $1.04 per unit, with disposal fees sometimes exceeding $15 per unit
Conclusion
Compliance and proper storage for wellness products isn't bureaucratic overhead—it's what protects product quality, consumer safety, and brand reputation from warehouse to doorstep. With the US dietary supplements market expected to reach $76.9 billion by 2030, even a single regulatory misstep—recalled inventory, a failed audit, or a cold-chain breach—can wipe out margins at a scale that's hard to recover from.
The right approach ties together compliant storage, disciplined inventory management, and a shipping setup matched to each product's specific needs. For most growing brands, that means working with a 3PL that understands wellness and medical product requirements from the ground up.
Bluebonnet Medical Supplies' FDA, GMP, and ISO-compliant operations cover exactly that:
- Purpose-built warehouse facilities with environmental monitoring
- Trained personnel who understand regulatory requirements
- FDA-cleared packaging with tamper-evident seals
- Established carrier relationships that reduce shipping costs while maintaining cold-chain integrity
Frequently Asked Questions
What FDA regulations apply to shipping dietary supplements in the US?
Dietary supplements fall under DSHEA and must meet FDA's 21 CFR Part 111 GMP standards, covering manufacturing, storage, packaging, and labeling—not just production. Warehouses and 3PLs are explicitly not exempt from these holding requirements.
Do wellness products require temperature-controlled shipping?
It depends on the product type. Most capsules and tablets can ship at controlled room temperature (59–77°F), while probiotics, some gummies, and functional beverages require cold-chain handling (36–46°F refrigerated) to preserve potency and shelf stability throughout transit.
What are the labeling requirements for wellness products sold online in the US?
Labels must include a Supplement Facts panel, ingredient list, net quantity, manufacturer/distributor contact information, and the FDA disclaimer for structure/function claims: "This statement has not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure, or prevent any disease."
How should probiotics be stored in a warehouse?
Probiotics typically require refrigerated storage at 2–8°C (35–46°F), humidity control below 40% RH, and protection from light. FEFO (First-Expire-First-Out) rotation should be applied to ensure the freshest stock ships first.
What happens if wellness products are shipped without proper compliance documentation?
Potential consequences include FDA warning letters, marketplace delisting (especially on Amazon), product seizure at customs or distribution points, and inability to execute a targeted recall if a quality issue arises. Industry estimates put direct recall costs in the millions, making preventive compliance far less expensive than the alternative.
What should I look for in a 3PL to handle wellness product fulfillment?
Prioritize GMP-certified operations with FDA-compliant storage, lot-level FEFO inventory tracking, and continuous temperature and humidity monitoring with documented records. Experience with supplements or medical products specifically matters more than general e-commerce volume.


