
The challenge goes beyond finding customers. Foreign wearable brands must navigate import duties, customs clearance, FDA compliance for medical-grade devices, and fulfillment logistics that meet the two-day delivery expectations of 74% of US online shoppers. Both consumer wearables (smartwatches, fitness trackers, earbuds) and medical-grade wearables (ECG monitors, continuous glucose monitors, clinical health trackers) face distinct but overlapping challenges when entering US distribution.
This article covers the distribution models available to foreign wearable brands, the regulatory landscape they must navigate, customs and import logistics strategies, and how brands are solving the fulfillment challenge with US-based partners.
TLDR:
- The US wearables market reached $19.92 billion in 2023 and is projected to hit $47.51 billion by 2030
- Cross-border DTC shipping is no longer viable due to eliminated de minimis exemptions and Section 301 tariffs
- Most scaling brands use US-based 3PLs to position inventory domestically, cutting delivery times and shipping costs by 15-30%
- Medical wearables require FDA-compliant storage and packaging—not all 3PLs are qualified
- Hybrid fulfillment models (3PL + Amazon FBA) provide the best balance of control and reach
The US Wearables Market: Why It's Worth the Complexity
The US wearables market presents a massive revenue opportunity that justifies navigating steep regulatory and logistical barriers. The US wearable technology market was valued at $19.92 billion in 2023 and is projected to grow at a 12.8% compound annual growth rate (CAGR) through 2030, reaching $47.51 billion.
Global wearable device shipments grew 9.1% year-over-year in 2025, reaching 611.5 million units. This growth is concentrated in specific categories:
- Wristbands: 14.7% growth in 2025
- Earwear: 7.8% growth globally
- Emerging form factors: Smart rings and display-less smart glasses gaining traction

Why the US Market Commands Premium Pricing
The US offers significantly higher per-device spending compared to emerging markets. India's wearable market is growing fast — 17.7% CAGR — but its average selling price (ASP) was just $19.20 in Q2 2025. The US market supports far higher price points for advanced health and fitness features, making it the primary target for brands with clinical-grade monitoring technology.
Brands from India, China, and Southeast Asia are increasingly chasing that premium. Smartwatches, health-monitoring wearables, and true wireless earbuds are driving the fastest US growth — but capturing that demand means navigating a distribution infrastructure that's far more complex than the home market.
Distribution Models Foreign Wearable Brands Use to Enter the US
Foreign brands entering the US must choose between several distribution models, each with distinct trade-offs in cost, control, and scalability.
Direct-to-Consumer (DTC) Cross-Border Model
Brands ship directly from overseas to US customers via international couriers like DHL or FedEx. This model works at low volumes but becomes costly, slow, and unpredictable at scale.
Key limitations:
- Ocean freight from Shanghai to Los Angeles takes 27-36 days
- The US eliminated the $800 de minimis exemption for Chinese-origin goods, exposing individual parcels to Section 301 tariffs and fees
- 74% of US online shoppers expect delivery within two days—cross-border shipping can't compete
Amazon FBA (Fulfillment by Amazon) Model
Brands ship inventory in bulk to Amazon's US warehouses, which handles storage, picking, packing, and last-mile delivery.
Advantages:
- Prime eligibility drives higher conversion rates
- Access to Amazon's massive customer base
- 86% of top Amazon sellers use FBA
Limitations:
- Fulfillment fees increased by $0.08 per unit for standard-size products priced $10-$50 in January 2026
- Storage fees fluctuate from $0.78/cubic foot (Jan-Sep) to $2.40/cubic foot (Oct-Dec)
- Amazon ended inventory commingling on March 31, 2026—brands now need FNSKU barcode stickers
- Restricted product categories and limited control over packaging and brand experience
US Wholesale/Distributor Model
Brands partner with an American distributor or retailer who takes on inventory risk and sells through their own channels (retail stores, online marketplaces, B2B networks).
Trade-offs:
- Reduces operational burden—no need to manage warehousing or fulfillment
- Significantly reduces margin (distributors typically take 30-50% markup)
- Loss of control over brand experience, pricing, and customer relationships
- Best suited for brands prioritizing market entry speed over margin preservation
Third-Party Logistics (3PL) Model
Unlike the distributor model—where you hand off control along with the inventory—a 3PL keeps you in the driver's seat. The brand ships bulk inventory to a US-based 3PL warehouse, which stores, picks, packs, and ships on their behalf. For brands scaling beyond early DTC volume, this has become the go-to model.
Why brands prefer this model:
- 3PLs achieve 15-30% shipping cost savings through negotiated carrier rates
- Full control over packaging, branding, and customer experience
- Scalable infrastructure without capital investment in US facilities
- Multi-channel fulfillment (DTC website, Amazon seller-fulfilled, retail orders)
Hybrid Model: The Emerging Standard
Many brands combine both: Amazon FBA handles visibility and Prime eligibility, while a 3PL processes direct website orders. The channel split typically looks like this:
- Amazon FBA: Standard SKUs, high-velocity products, price-sensitive buyers
- 3PL (DTC website): Custom packaging, bundled kits, compliance-sensitive items
- 3PL (retail/B2B): Wholesale purchase orders, retailer-specific labeling requirements
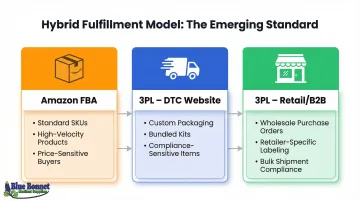
This structure lets brands capture Amazon's reach without surrendering packaging control or regulatory handling on every order.
Customs, Tariffs, and Import Logistics: What Brands Must Plan For
Importing wearable electronics requires precise Harmonized Tariff Schedule (HTS) classification. Misclassification leads to customs delays and unexpected duty bills.
HTS Classification and Duty Rates
Smartwatches are generally classified under HTS subheading 8517.62.00, which carries a Column 1 General duty rate of "Free." However, Chinese-manufactured devices are subject to Section 301 tariffs ranging from 7.5% to 25%.
Tariff impact by category:
| Wearable Category | Primary HTS Code | Base Duty (Column 1) | Section 301 Impact (China Origin) |
|---|---|---|---|
| Smartwatches | 8517.62.00 | Free | 7.5% - 25% additional tariffs |
| Patient Monitors (Medical) | 9018.19.55 | Free | Subject to Section 301 tariffs |
The Role of a US Customs Broker
Foreign brands shipping in commercial volumes need a licensed US Customs Broker to navigate entry documentation and compliance. Broker fees typically range from $125 to $175 for a formal ocean entry.
Required documentation:
- Commercial invoice
- Packing list
- Bill of lading
- Product certifications (FDA clearance, FCC compliance, etc.)
- Evidence of right to make entry
Additional fees:
First-Mile Shipping Strategy
Brands typically consolidate shipments at origin, ship ocean or air freight to a US port of entry, then clear customs before forwarding to a domestic warehouse. Those timeline and cost variables make the choice of freight mode one of the most consequential decisions in the inbound supply chain.
Timeline planning:
- Ocean freight from Shanghai to Los Angeles: 27-36 days for Full Container Load (FCL)
- Normal customs clearance: 24-48 hours
- Intensive customs exams at congested ports: 10-20 days
Air freight cuts transit time to 5-7 days but costs 4-6x more than ocean freight. Brands that carry high-margin, time-sensitive inventory typically absorb that premium; those with bulkier, lower-margin products generally commit to ocean freight and plan inventory buffers accordingly.

FDA and Regulatory Compliance for US-Bound Wearables
The FDA strictly regulates wearable devices that cross the line from "general wellness" to medical diagnostics.
The Wellness vs. Medical Device Distinction
Under the FDA's General Wellness Policy, products are exempt from medical device regulations if they are intended for general wellness use and present low risk to safety. General wellness claims relate to weight management, physical fitness, relaxation, or sleep management—without reference to diseases or conditions.
Products are NOT general wellness devices when they:
- Measure, estimate, or report physiologic values for medical or clinical purposes
- Are intended for screening, diagnosis, monitoring, alerting, or management of a disease or condition
FDA Enforcement: Recent Warning Letters
On July 14, 2025, the FDA issued a Warning Letter to Whoop, Inc., stating that its "Blood Pressure Insights" feature was an adulterated and misbranded medical device because it was intended for diagnosis of hypo- and hypertension without FDA clearance.
On February 21, 2024, the FDA warned consumers not to use smartwatches or smart rings that claim to measure blood glucose levels without piercing the skin, noting that no such devices have been authorized.
Regulatory Pathways for Medical Wearables
| Wearable Feature | FDA Classification | Regulatory Pathway Example |
|---|---|---|
| ECG / AFib Detection | Class II (Product Code: QDA) | Apple Watch ECG App (De Novo: DEN180044) |
| Continuous Glucose Monitoring (CGM) | Class II (Product Code: QBJ) | Dexcom G7 (510(k): K213919) |
FDA-Compliant Storage and Packaging Requirements
Clearing the regulatory pathway is only part of the equation. Once a device is authorized for sale, distribution infrastructure must meet the same compliance standards as the product itself.
Medical-grade wearables require FDA-compliant packaging and GMP-compliant storage. This means:
- Storage (21 CFR 820.150): Temperature controls for sensitive components, documented inventory systems, and procedures to prevent mixups or deterioration
- Labeling (21 CFR Part 801): Labels must name the manufacturer, packer, or distributor; all claims must align with FDA clearance scope; instructions for use must be clear and accurate
- Sterile packaging (when required): FDA recognizes ISO 11607-1 and ISO 11607-2 for terminally sterilized devices; packaging must maintain sterility until point of use
Consequences of Non-Compliance at the Distribution Level
Regulatory compliance carries through every stage of the supply chain, not just product launch. Non-compliant storage or packing at a fulfillment center can:
- Result in products being detained at customs
- Trigger FDA warning letters
- Lead to costly recalls
- Jeopardize a product's regulatory standing
Brands entering the medical wearable space need a US fulfillment partner whose infrastructure meets FDA, ISO, and GMP standards—not a general logistics warehouse. Bluebonnet Medical Supplies, a Cedar Park, TX-based medical 3PL, holds FDA-cleared medical packaging services along with ISO and GMP compliance, providing the specialized handling that regulated wearables require.
Why International Wearable Brands Partner with a US-Based 3PL
Rather than managing US warehousing, carrier relationships, and fulfillment staff from overseas, brands offload the domestic logistics layer to a specialized 3PL. This removes the need to establish a US entity, hire locally, or manage lease agreements.
Core Operational Benefits
What a quality US-based 3PL offers:
- Defers customs duties through bonded warehousing until products are sold, reducing upfront cash requirements
- Handles pick-and-pack with custom boxes, inserts, kitting, and protective materials suited to electronics
- Fulfills DTC, Amazon seller-fulfilled, and wholesale orders from a single shared inventory pool
- Provides 15-30% shipping cost savings through negotiated carrier rates that individual shippers can't access
The US third-party logistics market was valued at $247.4 billion in 2023 and is expected to grow at a 9.2% CAGR through 2030, driven by brands seeking scalable infrastructure without capital investment. For medical wearable brands specifically, not every 3PL can support these requirements.
Why Medical Wearable Brands Need Specialized 3PLs
A general e-commerce warehouse is not equipped for:
- HIPAA-safe data handling
- FDA-compliant labeling and packaging
- GMP-compliant storage with temperature controls
- Proper handling of sensitive electronic medical devices
Bluebonnet Medical Supplies, for example, operates with FDA-cleared medical packaging, ISO compliance, and GMP-compliant storage — meaning medical wearables maintain their regulatory standing from receiving through final-mile delivery.
Returns Handling: A Critical Domestic Capability
A US-based 3PL receives, inspects, restores, and either re-stocks or disposes of returned units domestically. For a brand operating from abroad, this is operationally essential — international returns shipping is prohibitively expensive and slow.
Returns workflow for wearables:
- Customer initiates return through brand's website or Amazon
- 3PL receives returned unit at US warehouse
- Unit is inspected and tested for functionality
- Defective units are assessed for warranty claims
- Functional units are restored (cleaned, repackaged) and returned to inventory
- Non-restorable units are properly disposed of or sent for refurbishment
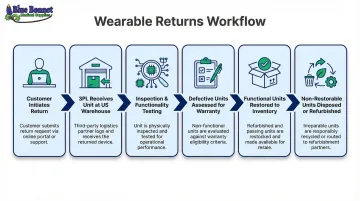
For medical wearables, returned devices may need to be de-labeled, re-inspected per FDA standards, or destroyed. Brands need a partner with documented processes for each scenario. Bluebonnet's product testing and restoration services are built specifically around these requirements.
Strategic Advantage: Inventory Positioning
Having stock already inside the US means:
- Meets two-day delivery expectations and keeps pace with domestic competitors
- Clears customs once per bulk shipment rather than on every individual order
- Responds to demand spikes without waiting on 4-6 week ocean freight cycles
- Supports flash sales, holiday promotions, and influencer campaigns with inventory that's ready to ship
Managing Inventory and Returns Across Borders
Inventory Forecasting for Long Lead Times
Foreign brands supplying a US 3PL must plan for longer lead times. Ocean freight from Asia takes 4-6 weeks, requiring brands to:
- Maintain safety stock to cover demand during transit
- Use inventory management tools that sync with the 3PL's warehouse management system (WMS)
- Plan seasonal demand 8-10 weeks in advance (accounting for production, transit, and customs clearance)
Best practices:
- Implement rolling 90-day demand forecasts
- Use historical sales data to predict seasonal spikes
- Communicate promotional plans to your 3PL 6-8 weeks in advance
- Monitor inventory levels weekly through WMS integration
Reverse Logistics: Handling Returns Domestically
Returned wearables need to be tested, assessed for defect vs. customer error, restored to resalable condition, and re-entered into inventory. A 3PL with product testing and restoration capabilities handles this without shipping units back to the country of origin.
Financial implications:
- Domestic return shipping: $5-$8 per unit
- International return shipping: $25-$40 per unit
- Restoration cost (cleaning, repackaging): $3-$5 per unit
- Disposal cost (non-restorable units): $2-$3 per unit
By handling returns domestically, brands save $20-$35 per returned unit while maintaining faster turnaround times.
Compliance Implications for Returned Medical Wearables
- Each returned medical wearable must be logged and assessed
- Defect patterns must be tracked and reported
- Returned devices may need to be quarantined pending investigation
- Some returned units may require destruction rather than restocking
Bluebonnet Medical Supplies manages the full cycle — quarantine, re-inspection, and device disposition — keeping clients aligned with FDA Quality System Regulations without the burden of coordinating it internally.
Frequently Asked Questions
How big is the US wearables market?
The US wearable technology market was valued at $19.92 billion in 2023 and is projected to reach $47.51 billion by 2030, growing at a 12.8% CAGR. Global wearable shipments reached 611.5 million units in 2025, growing 9.1% year-over-year.
Who are the leaders in wearable technology?
As of Q4 2025, Apple leads with 21.0% market share, followed by Xiaomi (10.3%), Huawei (8.0%), and Samsung (7.7%). Emerging brands from India and China are gaining ground in the affordable health-tracking segment.
What is the size of wearables market in India?
India's wearable technology market generated $3,708.7 million in 2025 and is expected to reach $13,556.9 million by 2033, growing at a 17.7% CAGR. However, the market is saturated with entry-level devices, with an average selling price of just $19.20 in Q2 2025.
Do wearable devices need FDA approval to be sold in the US?
Consumer fitness wearables don't require FDA clearance when limited to general wellness claims like fitness tracking or sleep monitoring. Wearables that measure clinical parameters (ECG, blood glucose, blood pressure) or make diagnostic claims are classified as medical devices and typically require 510(k) clearance or De Novo authorization before US sale.
What is the best fulfillment model for a foreign wearable brand entering the US?
Most scaling brands use a US-based 3PL, alone or alongside Amazon FBA, to position inventory domestically and cut delivery times to 1-2 days. This model delivers 15-30% shipping cost savings through negotiated carrier rates while keeping full control over packaging and returns.
Can a 3PL handle medical-grade wearable devices?
Not all 3PLs are qualified for this. Brands need a partner with FDA-compliant packaging credentials, GMP-compliant storage, and hands-on experience with regulated medical products. Bluebonnet Medical Supplies holds FDA-cleared medical packaging, ISO compliance, and GMP-compliant storage certifications, with operations built around medical device distribution.


