
Introduction
Indian DTC brands have proven their model domestically. Brands in beauty, wellness, health, and consumer goods are now eyeing the US as the natural next growth frontier. According to McKinsey & Company, India's D2C channel currently generates $10–12 billion in e-commerce sales and is projected to reach $60 billion by 2030. This ambition to cross borders is backed by real momentum, not just aspiration.
Most Indian DTC founders know how to build a product and market it. But the moment they try to serve a US customer fast, cheaply, and compliantly, they hit a wall. Fulfillment is where global ambitions stall.
Shipping directly from India creates slow delivery, high costs, and customs complexity. US-based inventory is the answer, but choosing the right fulfillment model, navigating FDA compliance, and selecting a logistics partner requires careful strategy.
This post unpacks why the US market is worth pursuing, what makes cross-border fulfillment genuinely hard, which fulfillment models work, what compliance traps to avoid, and how to evaluate a US logistics partner before committing.
TLDR
- Indian DTC brands are moving into the US market, pushed by diaspora demand, rising domestic competition, and stronger dollar margins
- US-based inventory solves the slow delivery and customs complexity that direct-from-India shipping creates
- Three fulfillment models dominate: Amazon FBA US, dedicated 3PL, and hybrid — and the right choice depends on your product type, volume, and compliance needs
- Health and medical product brands face strict FDA regulations including facility registration and labeling compliance
- The fulfillment partner you choose — their infrastructure, compliance credentials, and flexibility — determines whether your US launch succeeds or stalls
Why the US Is the Next Big Market for Indian DTC Brands
The case for Indian DTC expansion into the US comes down to two things: a diaspora that already buys your products and a domestic market that's getting harder to grow in.
The Diaspora Advantage
The US Indian-American population reached 5.2 million in 2023, making it the second-largest Asian origin group in the country. This demographic wields exceptional purchasing power, with a median household income of $151,200—nearly double the US average. For Indian brands in Ayurveda, wellness, and heritage-inspired beauty, this diaspora represents a built-in customer base that already understands and values the products.
The US Ayurveda market alone generated $1.54 billion in 2024 and is projected to reach $3.95 billion by 2030, demonstrating mainstream appetite beyond just the diaspora community.
Push Factors from the Domestic Market
India's domestic market is getting harder to scale profitably. Three pressures are pushing founders toward geographic diversification:
- Rising customer acquisition costs as paid channels become more competitive
- Heavy marketplace competition from entrenched domestic incumbents
- Investor and board pressure to demonstrate unit economics, not just growth
Rather than spending more to win the same customers at home, brands are finding better returns by entering underserved US niches early.
Proven Success Stories
Several Indian DTC brands have already gained US traction. WOW Skin Science launched in the US in 2018 and now sells in over 2,850 Walmart locations. Forest Essentials secured investment from The Estée Lauder Companies, which announced in 2026 it would acquire the remaining interests to expand the brand globally. Both entered the US before their respective categories hit mainstream awareness—which is exactly where several Ayurveda and wellness verticals sit right now.
The Real Fulfillment Challenges of Cross-Border Expansion to the US
The "Ship from India" Trap
Some brands start by fulfilling every US order from India. It fails fast. Delivery times of 10–20 days are unacceptable against expectations shaped by Amazon Prime.
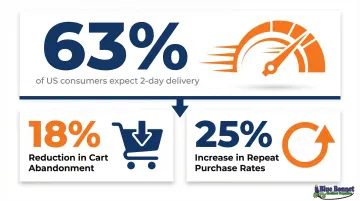
63% of US consumers expect two-day delivery, and offering it can reduce cart abandonment by 18% and increase repeat purchase rates by 25%.
Cost per shipment is high, tracking is inconsistent, and customs delays can stall entire order batches at the port.
The End of Section 321 De Minimis
The economics got worse in 2025. Executive Order 14324 suspended the $800 duty-free exemption for all countries effective August 29, 2025. Low-value shipments now face specific duties or ad valorem rates, breaking the unit economics of direct-from-India B2C fulfillment.
Customs and Duties Complexity
Imports into the US require accurate HTS (Harmonized Tariff Schedule) codes, proper commercial invoices, and correct declared values. Errors mean shipments held, fines, or returned goods. De minimis thresholds are no longer a sustainable fulfillment strategy for scaling brands.
Inventory Commitment Risk
Moving stock into a US warehouse requires forecasting demand in an unfamiliar market. Overstocking ties up working capital; understocking means stockouts and lost customers. This inventory uncertainty is one of the biggest hesitations Indian founders have about committing to US fulfillment infrastructure.
The Returns Problem
US consumers return products at significantly higher rates than Indian consumers. In 2023, the US online return rate was 17.6%, accounting for $247 billion in merchandise. They also expect fast, frictionless returns.
Without a US-based return processing point, every return becomes an international logistics event — expensive, slow, and operationally painful. Brands that can't handle returns locally will see repeat purchase rates suffer.
Operational Bandwidth Gap
Brands that are still founder-led or lean on ops often lack the bandwidth to manage a second logistics geography. A new carrier network, new fulfillment SLAs, and a different timezone all run in parallel with the India business. That operational load rarely shows up in early expansion models — until it becomes a problem.
US Fulfillment Models: FBA, 3PL, or Hybrid
Amazon FBA US: The Fast Entry Point
Many Indian brands start with Amazon FBA US because it gives instant access to Prime delivery, trusted logistics infrastructure, and a built-in customer base. In 2025, approximately 82% of active Amazon marketplace sellers utilized FBA.
Ship inventory to an Amazon fulfillment center in the US, list on Amazon.com, and FBA handles picking, packing, delivery, and returns. The trade-off is real, though: FBA locks you into Amazon's channel and fees, limits branding, and restricts fulfillment from your own website or other US platforms.
2026 FBA costs to watch:
- Fulfillment fees increased by an average of $0.08 per unit
- Storage costs surge 208% during Q4 peak season (October–December)
- Inbound defect fees range from $0.32 to $5.72 per unit for mislabeled shipments
Dedicated US 3PL: Multi-Channel Control
A third-party logistics provider stores your inventory in a US warehouse and fulfills orders across all your sales channels — your DTC website, Amazon, Walmart, and more. You get control over packaging, brand experience, and returns handling. This is the preferred model for brands growing beyond Amazon-only and building direct relationships with US customers.
For health and medical product brands specifically: Working with a compliant 3PL—one with FDA-cleared packaging capabilities, ISO and GMP compliance—is not optional; it's a regulatory necessity. Bluebonnet Medical Supplies, for example, operates with FDA-cleared medical packaging, ISO and GMP compliance, and specialized capabilities for handling sensitive health products. When evaluating 3PL partners for medical products, these credentials are the floor, not a bonus.
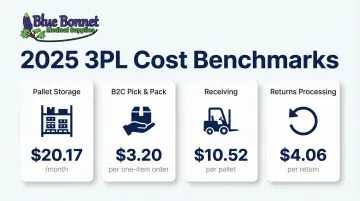
Typical 3PL costs (2025 benchmarks):
| Service | Average Cost |
|---|---|
| Pallet storage | $20.17/month |
| B2C pick and pack | $3.20 per one-item order |
| Receiving | $10.52 per pallet |
| Returns processing | $4.06 per return |
Hybrid Approach: FBA + 3PL Running in Parallel
Many brands run FBA for Amazon marketplace velocity while using a 3PL for their Shopify/DTC website orders. This separates inventory, which adds complexity, but allows brands to serve both channels effectively.
- When this makes sense: Higher order volume, established US demand, budget for dual inventory commitment
- When it adds overhead: Early-stage testing, limited inventory, tight cash flow
Freight Forwarding + Bonded Warehouse for Testing
Before committing to any of the above models, some brands ship a test batch via freight forwarder into a bonded warehouse or small US storage facility to validate demand. It works as a lower-risk proof-of-concept, but won't scale for live order fulfillment.
Model Comparison
| Model | Cost Structure | Delivery Speed | Channel Flexibility | Compliance Capability | Branding Control |
|---|---|---|---|---|---|
| Amazon FBA | Per-unit fees + storage | 2-day Prime | Amazon only | Basic | Low |
| Dedicated 3PL | Monthly + per-order | 2-3 days | All channels | High (if specialized) | High |
| Hybrid | Dual fees | 2-3 days | All channels | High | High |
| Freight Forwarding | Per-shipment | Testing only | N/A | Varies | N/A |
US Compliance and Regulatory Requirements for Indian Brands
FDA Requirements for Health, Wellness, and Medical Products
This is the highest-stakes compliance area for Indian brands in supplements, Ayurvedic formulations, skincare with drug claims, and medical devices.
Key FDA requirements:
- Foreign manufacturers must comply with 21 CFR Part 111 Current Good Manufacturing Practices, which requires establishing specifications and at least one test to verify the identity of any dietary ingredient
- FDA Import Alert 54-14 places non-compliant foreign supplement manufacturers on a Red List, subjecting their products to Detention Without Physical Examination (DWPE) and automatic refusal of entry
- Manufacturers must submit a premarket notification to the FDA at least 75 days before introducing a dietary supplement containing a New Dietary Ingredient

Skipping FDA compliance means shipments get detained at the border. For a brand timing a US product launch, that's an expensive way to learn the rules.
US Customs Import Requirements
Every shipment entering the US must have:
- Accurate HTS code
- Proper country of origin labeling
- Valid commercial invoice
- CPSC (Consumer Product Safety Commission) certification or other product safety documentation for certain categories
Requirements vary by product category — textiles, cosmetics, food supplements, and medical devices each carry distinct documentation rules. Know your checklist before the first shipment leaves India.
Product Labeling Compliance
US labeling rules differ significantly from India:
- English-only labeling required
- Net weight must be in US customary units as well as metric
- Ingredient lists follow FDA sequence rules
- Any health or structure-function claims must meet FTC/FDA standards
Non-compliant labels mean US Customs will reject the entire import.
The Compliance Advantage of a Specialized 3PL
Managing these requirements in-house adds overhead most Indian DTC brands aren't equipped for yet. A 3PL that already holds the right credentials handles storage, handling, and shipping in full compliance from day one.
When evaluating partners, look for:
- FDA-cleared medical packaging services
- ISO compliance
- GMP compliance
Bluebonnet Medical Supplies carries all three, with a warehouse specifically built to handle sensitive medical and health products.
How to Choose a US Fulfillment Partner
Baseline Requirements
A US 3PL must meet these criteria for Indian DTC brands:
- Geographic coverage: Single warehouse vs. bicoastal strategy affects delivery speed. A bicoastal strategy reduces shipping zones and lowers carrier costs while achieving 2-day delivery to a broader customer base
- Technology integration: Can they connect with Shopify, Amazon, WooCommerce?
- Transparent pricing: Per-unit pricing, no hidden fees
- Returns processing: Do they offer product inspection and restocking services?

Category Fit Over Price
Baseline requirements get you in the door—but category fit determines whether the partnership actually works. A general-purpose warehouse that handles furniture and apparel may not have the temperature control, handling protocols, or compliance credentials needed for sensitive health or medical products.
Screen potential partners with these questions:
- What experience does the 3PL have with your product category?
- What compliance certifications are relevant to that category?
- Do they offer custom packing solutions or just a one-size-fits-all system?
For medical and health products, this matters more than cost. A 3PL like Bluebonnet Medical Supplies, which holds FDA, ISO, and GMP compliance credentials and handles products to regulatory standards, removes the burden of verifying every operational detail yourself.
Hidden Costs Founders Miss
- Storage fees that compound if inventory moves slowly
- Pick-and-pack charges per unit
- Special handling surcharges
- Inbound receiving fees
- Return processing costs
Model your full landed cost (manufacturing + freight to US + duties + 3PL fees + last-mile carrier) before committing to a fulfillment model. While you're at it, ask whether the 3PL passes carrier discount rates on to clients—this alone can meaningfully reduce last-mile costs.
Onboarding Timeline
A well-executed 3PL onboarding process typically takes 4 to 8 weeks from contract signing to full launch. This includes discovery, systems integration (connecting Shopify/ERP to the WMS), workflow design, and pilot testing. Plan your US launch timeline accordingly.
Frequently Asked Questions
Do Indian DTC brands need a US business entity to use a US fulfillment warehouse?
While a foreign company can export to the US without a US entity, forming a US corporation or LLC is highly recommended. It shields the parent company from liability, simplifies acting as the Importer of Record, and is often required to open US bank accounts and secure domestic payment processors.
What US customs duties apply to Indian DTC brands shipping products into the US?
Duties are determined by the HTS code for your product category and country of origin. Founders should research the applicable duty rate for their specific product type, noting that some categories (such as certain textiles or processed foods) face higher rates than others.
How long does it realistically take to set up US-based fulfillment operations?
From selecting a 3PL partner and onboarding (2-4 weeks) to shipping the first inbound inventory to the US warehouse (4-8 weeks including freight transit), founders should plan for a 6-12 week timeline to launch US fulfillment operations.
Should Indian DTC brands use Amazon FBA US or a dedicated 3PL for their first US fulfillment setup?
FBA is a faster, lower-barrier starting point for brands focused on Amazon US, while a dedicated 3PL is better for brands that want to sell across multiple channels including their own website. Many brands eventually use both in a hybrid model.
What specific FDA requirements apply to Indian health and wellness brands selling in the US?
Key requirements include facility registration for food/supplement manufacturers, labeling compliance (Supplement Facts, NDI notifications for new ingredients), and awareness of import alert risks. Founders should consult a US regulatory specialist before their first shipment.
How do Indian DTC brands manage product returns from US customers cost-effectively?
Working with a US-based 3PL that offers in-country returns processing is the most practical option. This allows returns to be inspected, restocked, or disposed of without sending products back to India, protecting margins and customer experience.


