
Introduction
Medical product businesses are turning to Amazon FBA for good reason: more than 600 active U.S. fulfillment facilities and a global e-commerce medical device market valued at $25 billion. The reach is real. So are the compliance requirements that come with it.
FBA is not plug-and-play for medical products. FDA compliance must be in place before a single unit ships to an Amazon warehouse.
The requirements are specific and enforceable:
- FDA facility registration with an active FEI number
- Accurate labeling under 21 CFR Part 801
- Proper Importer of Record designation through CBP Form 5106
- Amazon's own medical device listing policies
Miss any one of these and the consequences follow quickly: cargo detained at the port, listings removed without notice, or permanent customs refusal under the "appears to violate" legal standard.
TLDR
- Amazon FBA medical products must meet both FDA import rules and Amazon's device policies
- FDA facility registration (FEI number) and U.S. Importer of Record status are required before shipment—Amazon does not fill this role
- Many device categories need Amazon pre-approval; verify eligibility before sending inventory
- Non-compliant labeling is the top reason FDA detains shipments at customs
- An FDA-compliant 3PL ensures products are prepped, documented, and FBA-ready before entering Amazon's network
How FBA Works for Medical Product Sellers: Opportunities and Compliance Realities
Amazon's FBA model is straightforward: sellers ship inventory to Amazon's fulfillment centers, and Amazon handles storage, picking, packing, shipping, and customer service. For medical product companies looking to scale without managing their own logistics, this is attractive. But that convenience comes with conditions.
Before a single unit ships to Amazon, sellers carry full FDA compliance responsibility. Key realities to understand:
- Amazon warehouses are not FDA-registered facilities for medical devices
- Amazon does not act as the Importer of Record
- The seller—or their designated U.S. entity—bears full compliance responsibility before and during shipment
Amazon operates more than 600 active fulfillment facilities across the United States, and sellers cannot choose which warehouse receives their inventory. This makes advance compliance documentation even more important, since products may enter through multiple ports and be routed to any facility in Amazon's network.
FDA Registration and Documentation Requirements for FBA Medical Product Shipments
FDA Facility Registration and FEI Number
Any facility that manufactures, processes, packs, or holds FDA-regulated medical devices for U.S. distribution must register with the FDA and obtain an FDA Establishment Identifier (FEI) Number under 21 CFR 807.20. This registration must be current before products ship to any Amazon warehouse.
According to 21 CFR Part 807, "No device may be imported or offered for import into the United States unless it is the subject of a device listing and is manufactured, prepared, propagated, compounded, or processed at a registered foreign establishment." Foreign establishments without active registration will have their shipments refused at the port.
Importer of Record (IOR) and CBP Form 5106
The foreign exporter—not Amazon—must serve as the U.S. Importer of Record. This requires obtaining a U.S. Customs Assigned Number through CBP Form 5106, through customs brokers like FedEx, UPS, or DHL.
What the IOR number is used for:
- Bond coverage
- Entry processing
- Liquidation
- Refund issuance
CBP defines the Importer as "the person primarily liable for the payment of any duties on the merchandise," which can be the consignee, importer of record, or actual owner (19 CFR 101.1).
Prior Notice and FSVP: Food Rules That Don't Apply to Devices
Prior Notice (21 CFR Part 1, Subpart I) and the Foreign Supplier Verification Program (FSVP, 21 CFR Part 1, Subpart L) apply exclusively to food and feed products—not medical devices. That said, carriers often can't make that distinction on their own.
Major carriers like FedEx, UPS, and DHL may still request documentation to confirm product category. Without clear classification paperwork, shipments can be delayed even when all other documentation is in order. Keep the following on hand to avoid carrier holdups:
- Product classification documentation (FDA device listing number)
- Commercial invoice with HS tariff code
- Certificate of FDA registration or exemption letter
FDA Detention: The "Appears to Violate" Standard
FDA detention means the agency or CBP holds an imported product at the port of entry because it "appears to violate U.S. laws and regulations" — a deliberately low bar under 21 U.S.C. § 381(a). The FDA doesn't need to prove a violation to detain a shipment; the appearance of one is enough.
In practice, detention results in:
- Delays of days or weeks at the port
- Increased costs for storage and demurrage (fees for delayed container return)
- Potential permanent refusal of entry if compliance cannot be demonstrated
- Destruction or return of goods at the importer's expense

Amazon's Medical Device Policies: What Sellers Need to Know
Amazon operates as its own regulatory layer on top of FDA rules. The platform restricts or requires pre-approval for many medical device categories, and sellers must confirm their product is eligible for FBA listing before shipping inventory to a fulfillment center.
Restricted and Pre-Approval Categories
Amazon requires sellers to provide FDA facility registration, 510(k) clearance (where applicable), and English labeling for Class II non-exempt devices. Categories that require Amazon approval before listing include:
- Asthma inhalers
- Defibrillators
- CPAP/BiPAP machines
- Surgical kits
- Ultrasound therapy devices
- Prescription (Rx) medical devices (prohibited from FBA entirely)
Prescription medical devices cannot be fulfilled via FBA — full stop. Amazon explicitly states: "Fulfillment by Amazon (FBA) shipments are prohibited. If an Rx medical device is shipped via FBA, it will be disposed of at the seller's expense." Rx devices must be sold through the Amazon Business Professional Healthcare (PHC) program and shipped via Merchant Fulfillment Network (MFN).
Product Listing Requirements and Health Claims
Amazon's algorithm flags and removes listings automatically—often without seller notice—when they violate health claim or approval language rules.
Prohibited listing content:
- Unsubstantiated health claims
- Disease-treatment language
- Language that implies FDA approval where none exists
- Use of the FDA logo in images or copy
Sellers cannot claim "FDA approved" for products that are only cleared or registered. Misuse of these terms triggers both Amazon takedowns and potential FDA enforcement action.
FDA-Cleared vs. FDA-Approved vs. FDA-Registered
Getting these terms right protects your listing — and keeps you out of FDA enforcement territory:
| Term | Meaning | Applicable Device Class | Can You Use It? |
|---|---|---|---|
| FDA Cleared (510(k)) | Device is substantially equivalent to a legally marketed predicate | Class I and II (non-exempt) | Yes, if you have 510(k) clearance |
| FDA Approved (PMA) | Device has undergone rigorous Premarket Approval demonstrating safety and efficacy | Class III (life-sustaining, implantable) | Yes, if you have PMA approval |
| FDA Registered | Facility is administratively enrolled in the FDA database | All classes | No—registration does not denote approval of products |
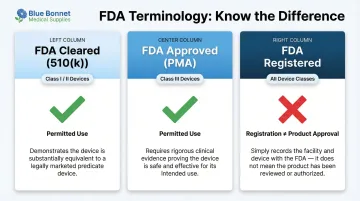
FDA explicitly states: "Registration of a device establishment, assignment of a registration number, or listing of a medical device does not in any way denote approval of the establishment or its products by FDA."
Category Application and Documentation Submission
Amazon requires documentation submission for category approval, including:
- 510(k) clearance letters
- Premarket approval (PMA) letters
- FDA facility registration confirmation
Submit incomplete documentation and Amazon may strand your inventory at the fulfillment center — or refuse the shipment entirely. Working with a compliance-experienced fulfillment partner ensures your paperwork is in order before inventory ships.
Common FDA Compliance Failures That Get Medical Products Detained or Removed
Shipping Without an Active FDA Registration
Products from facilities without a current FDA FEI registration will be flagged by CBP and refused entry. The shipment gets turned away entirely — and you're still on the hook for duties, storage fees, and re-export costs.
FDA's Data Dashboard tracks common refusal charges:
- NO 510(K): "The article is subject to refusal of admission in that it is a post-1976 device for which a Section 510(k) application does not appear to have been determined substantially equivalent."
- NOT LISTED: "It appears the drug or device is not included in a list required by Section 510(j)."
- DEVICE GMP: Methods or facilities do not conform to Good Manufacturing Practices under 21 CFR 820.
Non-Compliant or Missing Labeling
Registration gets your shipment through the door — but labeling errors can stop it at the same threshold. FDA-regulated medical devices must include specific labeling elements under 21 CFR Part 801:
- Device name
- Intended use
- Manufacturer name and address (including street, city, state, zip)
- Instructions for use
- Warning and precaution statements
- Expiration or manufacturing dates (format: YYYY-MM-DD)
- Unique Device Identifier (UDI) where applicable
Incorrect, missing, or foreign-language-only labels are among the most frequently cited reasons FDA issues detention notices at U.S. ports of entry.
Missing or Incorrect Product Codes and HTS Codes
Even properly labeled products with valid registrations can get held if the entry data is wrong. When submitting to CBP, sellers must provide accurate Harmonized Tariff Schedule (HTS) codes paired with the correct FDA product code — errors route the shipment to FDA review without supporting documentation, triggering automatic holds.
Common ACE data errors:
| Entry Field | Common Error | Root Cause | Corrective Action |
|---|---|---|---|
| DEV/DFE | "DEV/DFE did not match firms" | Firm name/address in ACE doesn't match FDA registration database exactly | Provide the FEI Number in ACE to force an exact database match |
| PM# | "PM# not transmitted and is req'd" | Product code requires a 510(k) or PMA, but the Affirmation of Compliance was omitted | Transmit PM# with correct qualifier (K, P, or DEN) |
| IUC | Manual Review Hold | Filer submitted "UNK" (Unknown) for Intended Use Code | Pre-clear the exact IUC (e.g., 081.001 for standard import of finished device) with your broker |

Attempting to Use Amazon's Address as the Importer of Record
Amazon's fulfillment centers cannot and do not accept IOR responsibility. Sellers who try to route customs documentation through Amazon's address will have entries rejected, which delays receipt of inventory and can result in Amazon canceling the inbound shipment.
Specific consequences include:
- CBP rejects the entry entirely, requiring re-filing under the correct IOR
- Amazon may cancel the inbound shipment before inventory is received
- Re-routing and re-filing fees add cost and delay that compound the original error
FDA-Compliant Labeling for Medical Products on Amazon
Key FDA Labeling Requirements (21 CFR Part 801)
Physical product labels must meet FDA requirements before shipment to FBA:
- Manufacturer or distributor identification (801.1): include name, street address, city, state, and zip code
- Adequate directions for use (801.5): written so a general user can operate the device safely for its intended purpose
- Standardized date format (801.18): expiration and manufacturing dates must follow YYYY-MM-DD (e.g., 2025-06-15)
- Unique Device Identifier (801.20): required on every device label and package; Class I devices are exempt from including a production identifier
FDA-Required Labels vs. Amazon Listing Content
These are two separate compliance requirements that must both be satisfied simultaneously:
- Physical product label must meet FDA requirements under 21 CFR Part 801
- Amazon product page must avoid disease claims or unapproved language per Amazon's medical device policies
Language and Multi-Language Labels (801.15)
Labeling must be in English for products sold in the U.S. market. Multi-language labels are acceptable as long as English requirements are fully met.
Per 21 CFR 801.15: "All words, statements, and other information required by or under authority of the act to appear on the label or labeling for a device shall appear thereon in one or more of the following formats: (A) The English language."
If the label contains any representation in a foreign language, all required information must also appear in that foreign language.
Review all labels before shipping to the FBA warehouse. Relabeling at the fulfillment center is expensive — and Amazon may reject non-compliant inventory outright.
How an FDA-Compliant 3PL Helps Medical Brands Scale on Amazon
Many medical product sellers underestimate the compliance burden between manufacturing and Amazon's fulfillment center. Products must be properly stored, packaged, labeled, and documented before they enter FBA—and a 3PL warehouse certified for medical products handles each of these pre-FBA steps, keeping sellers out of the regulatory weeds.
What an FDA-compliant 3PL provides:
- FDA-cleared packaging services with proper labeling, barcoding, and tamper-evident seals
- ISO and GMP compliance built into daily warehouse operations
- Organized storage systems designed for sensitive medical items
- Compliance checks to prevent customs delays
- Proper documentation for FDA and CBP entry requirements

Bluebonnet Medical Supplies, based in Cedar Park, Texas, is built specifically around these requirements. Beyond the standard pre-FBA checklist, they offer returns processing, product restoration for refurbished inventory, and direct carrier relationships that reduce shipping costs. Their model is deliberately non-corporate — each client gets a tailored logistics setup rather than a rigid, one-size-fits-all process. For medical brands scaling on Amazon, that flexibility matters as much as the compliance credentials.
Frequently Asked Questions
What is Amazon FDA?
"Amazon FDA" describes how Amazon's marketplace policies and FDA regulations overlap for sellers of regulated products. If you sell FDA-regulated items through FBA, you must meet both Amazon's listing requirements and FDA import and registration rules.
Is everything on Amazon FDA approved?
Not all products on Amazon are FDA approved or cleared. Amazon allows many FDA-regulated products to be sold, but sellers are responsible for ensuring their products meet required FDA standards. Amazon does not verify or guarantee FDA compliance on behalf of third-party sellers.
Do medical devices need FDA registration to sell on Amazon FBA?
Yes. Facilities manufacturing or distributing medical devices for the U.S. market must be registered with the FDA and have an active FEI number. Without this, shipments can be detained at customs and products cannot legally enter U.S. distribution.
What happens if my medical product is detained by FDA at customs?
The importer must respond to the detention notice with documentation proving compliance. If the product cannot be brought into compliance, it will be refused entry and returned or destroyed at the importer's expense.
Does Amazon require FDA clearance for medical devices sold via FBA?
Amazon requires sellers in restricted medical device categories to submit FDA clearance or approval documentation as part of the category approval process. Without this, listings may be blocked or removed, and inventory shipped to FBA may become stranded.
What documents do I need to ship FDA-regulated medical products to Amazon FBA?
You'll typically need the following:
- FDA facility registration (FEI number)
- U.S. Customs Assigned Number (CBP Form 5106 / IOR number)
- Bill of Lading or Airway Bill
- Commercial invoice
- FDA-compliant product labeling
- 510(k) clearance or PMA approval letter (where applicable)


