
Introduction
Choosing the wrong distribution partner costs medical product businesses more than time. It can mean failed regulatory inspections, customs holds, damaged goods, and customers who don't come back. The US medical supply distribution industry is a $502.5 billion sector—and for companies manufacturing or selling medical products, the right logistics partner directly determines whether products arrive on time, clear compliance checks, and reach customers without costly setbacks.
This guide is written for medical product companies—Amazon sellers, e-commerce brands, medical device manufacturers, and DTC healthcare businesses—who need a distribution partner built for regulated products, not just a warehouse that ships boxes. We'll cover the market landscape, profile the five largest US distributors, and outline a clear framework for evaluating partners on compliance credentials, handling capabilities, and service flexibility.
TLDR
- Medical supply distributors must meet FDA, ISO 13485, and GMP compliance for storage, packing, and shipping
- The Big 5 US distributors are McKesson, Cardinal Health, Medline, Henry Schein, and Owens & Minor
- Selection criteria include regulatory certifications, product handling infrastructure, shipping network reach, and workflow flexibility
- Small-to-mid-sized medical product businesses often need a personalized 3PL partner rather than enterprise-scale distributors
- Evaluate partners by capability and compliance fit, not just brand recognition or company size
Overview of Medical Supply Distribution in the US
Medical supply distribution is the intermediary function that moves healthcare products from manufacturers to end users—hospitals, clinics, pharmacies, and direct-to-patient channels—while managing compliant storage, regulatory documentation, and logistics operations.
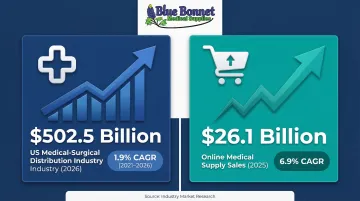
The US medical-surgical distribution industry reached $502.5 billion in 2026, growing at a 1.9% CAGR between 2021 and 2026. The sector spans 8,918 businesses focused on medical and surgical equipment, instruments, and supplies.
Online medical supply sales tell a separate story: $26.1 billion in 2025, expanding at a 6.9% CAGR as e-commerce adoption and direct-to-consumer healthcare models accelerate.
For medical product businesses, two distributor types matter most:
- Large enterprise distributors — serve healthcare facilities at scale with standardized, high-volume systems
- Specialized 3PL providers — serve product companies needing compliant storage, custom packing, and flexible shipping without enterprise-level volume commitments
Top US Medical Supply Distribution Companies
These companies were evaluated based on US market presence, regulatory compliance standing, breadth of services, and ability to serve diverse medical product businesses—from hospital networks to online sellers.
McKesson Corporation
McKesson has been operating since 1833, now headquartered in Irving, Texas, and ranked #9 on the Fortune 500. While primarily known for pharmaceutical distribution, its Medical-Surgical Solutions segment generated $11.4 billion in FY2025, serving over 340,000 customers across the alternate-site care spectrum.
What sets them apart: McKesson operates 50+ distribution centers offering 245,000+ national and private-brand products. The company specializes in serving physician offices, surgery centers, nursing homes, and home health agencies with comprehensive serialization solutions and DEA-registered controlled substance handling.
| Criteria | Details |
|---|---|
| Service Scope | Pharmaceuticals, medical-surgical supplies, health IT solutions, and specialty distribution across North America |
| Key Compliance / Certifications | DEA registration for controlled substances, FDA establishment registration, DSCSA serialization compliance for pharmaceuticals |
| Best Suited For | Large healthcare systems, hospital networks, and enterprise-level medical product companies needing national-scale distribution |
Cardinal Health
Cardinal Health, founded in 1971 and headquartered in Dublin, Ohio, ranks #15 on the Fortune 500. For FY2025, the company reported $222.6 billion in total revenue, with its Global Medical Products and Distribution (GMPD) segment contributing $12.6 billion.
Why it stands out: Cardinal Health operates as both a global manufacturer and distributor, supporting over 30 million surgical procedures annually. Recent expansions include new distribution centers in Walton Hills, Ohio, and Boylston, Massachusetts, strengthening their Northeast US footprint.
| Criteria | Details |
|---|---|
| Service Scope | Medical and surgical products, pharmaceutical distribution, lab products, and at-home patient care supplies |
| Key Compliance / Certifications | FDA establishment registration, DSCSA serialization services, UDI management through 3PL division |
| Best Suited For | Hospitals, pharmacies, specialty clinics, and manufacturers seeking integrated supply chain and distribution solutions |
Medline Industries
Headquartered in Northfield, Illinois, and founded in 1966, Medline completed a $6.26 billion IPO in February 2026. At $28.4 billion in net sales for FY2025, it stands among the most scaled manufacturer-distributors of medical supplies in the US.
Why it stands out: Medline runs one of the most vertically integrated operations in the industry. They offer approximately 335,000 products, roughly 190,000 of which are Medline-branded. Operating 69 global distribution centers and 26 North American manufacturing sites, they deliver next-day service to 95% of US customers while maintaining competitive private-label pricing.
| Criteria | Details |
|---|---|
| Service Scope | Clinical supplies, surgical products, PPE, rehabilitation equipment, and custom procedure trays |
| Key Compliance / Certifications | ISO 13485 certification, Medical Device Single Audit Program (MDSAP) certification, FDA establishment registration as specification developer and manufacturer |
| Best Suited For | Hospitals, long-term care facilities, and healthcare providers seeking a single-source supply partner with private-label options |
Henry Schein
Henry Schein (Fortune 500 Rank: 333) reported $13.2 billion in sales for 2025. Founded in 1932 and headquartered in Melville, New York, the company operates in 34 countries with a catalog of over 300,000 branded and corporate-brand products.
Why it stands out: Henry Schein specializes exclusively in office-based practitioners—primarily dental and medical offices. Beyond product distribution, they bundle technology solutions and practice management services, making them particularly valuable for specialty care settings that need integrated operational support.
| Criteria | Details |
|---|---|
| Service Scope | Medical, dental, and veterinary supplies, technology solutions, and practice management services for office-based practitioners |
| Key Compliance / Certifications | ISO 13485 certifications across multiple North American facilities |
| Best Suited For | Office-based physicians, dentists, veterinarians, and specialty practices needing a full-service distribution and practice management partner |
Owens & Minor
Owens & Minor has been operating since 1882, now headquartered in Richmond, Virginia, with $10.7 billion in net revenue for FY2024. Their 40+ distribution centers serve 4,000 healthcare partners with a focus on cost-effective supply chain solutions.
Why it stands out: Owens & Minor differentiates through its Patient Direct segment (including Byram Healthcare), providing direct-to-home delivery of medical supplies for chronic conditions like diabetes and ostomy care. They also offer proprietary supply chain consulting services (QSight) that help healthcare facilities reduce spend without compromising quality.
| Criteria | Details |
|---|---|
| Service Scope | Medical and surgical supply distribution, home healthcare products, patient-direct delivery, and supply chain consulting |
| Key Compliance / Certifications | FDA establishment registration, ISO certifications, HIPAA Business Associate Agreement capabilities for patient-direct fulfillment |
| Best Suited For | Healthcare facilities prioritizing cost efficiency, home health programs, and supply chain optimization partnerships |

How to Choose the Right Medical Supply Distribution Partner
The most common mistake businesses make is choosing a distributor based solely on brand reputation or revenue size—without evaluating whether the company's systems, compliance capabilities, and service model actually align with their specific product type, volume, and regulatory requirements.
Regulatory Compliance Is the Starting Point
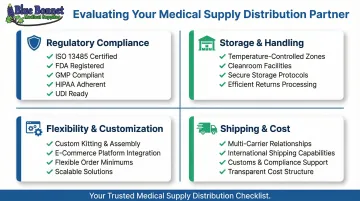
As of February 2, 2026, the FDA's new Quality Management System Regulation (QMSR) fully incorporates ISO 13485:2016 into 21 CFR Part 820. Any distributor handling packaging, relabeling, or storage of finished devices must demonstrate ISO 13485 certification as a baseline. Beyond that, look for:
- FDA establishment registration (for repackagers/relabelers)
- GMP compliance for storage and handling procedures
- HIPAA Business Associate Agreements (for patient-direct fulfillment)
- UDI traceability systems (for medical devices)
Improper handling or storage can lead to customs holds, product recalls, or regulatory penalties. Recent FDA enforcement—including a November 2024 Warning Letter to Owens & Minor for QSR violations and a 2022 McKesson recall linked to facility temperature excursions—proves that even Tier-1 distributors face severe operational lapses. Don't rely on brand name alone. Implement strict Quality Assurance Agreements (QAAs) that mandate real-time environmental monitoring and routine audits of the specific facilities housing your products.
Storage and Handling Capabilities
Sensitive medical items often require temperature-controlled, contamination-free environments. Confirm whether the warehouse is built to handle medical-grade inventory, not just general merchandise. Ask about:
- Temperature and humidity controls with real-time monitoring
- Cleanroom or contamination-controlled zones for sterile products
- Secure storage for high-value or controlled items
- Returns processing and product restoration capabilities
Flexibility and Customization
Large enterprise distributors operate at scale with standardized systems—which may not suit medical product companies with unique workflows, SKU complexity, or international shipping requirements. Look for a partner that can tailor logistics to your business model:
- Custom packaging and kitting services
- Integration with e-commerce platforms (Amazon FBA, Shopify, WooCommerce)
- Flexible order minimums and scalable pricing
- Direct communication with account managers, not automated systems
Shipping Network and Cost Structure
The right distributor should offer more than basic delivery. Key factors to evaluate:
- Direct carrier relationships with negotiated shipping rates
- International shipping capabilities and customs compliance support
- Returns processing and product restoration for resalable items
Finding the Right Fit for Your Business Size
For small-to-mid-sized medical product businesses—Amazon sellers, DTC brands, and device manufacturers who need full compliance without enterprise-level volume—a specialized 3PL partner is often a stronger match than a national distributor. Companies like Bluebonnet Medical Supplies (FDA-cleared, ISO and GMP compliant, HIPAA-safe handling) offer custom logistics workflows, direct account access, and compliance rigor built around your specific product requirements rather than a generic template.
Conclusion
The best medical supply distribution partner isn't the biggest name—it's the one that fits your actual products, workflows, and compliance requirements. When evaluating options, focus on what matters:
- Regulatory certifications: ISO 13485, FDA registration, GMP compliance
- Handling infrastructure: temperature control, cleanroom conditions, secure storage
- Operational fit: ability to scale with your business or adapt to your processes
If you're a medical product business looking for a compliant, personalized distribution partner that treats your business as more than just another account, reach out to Bluebonnet Medical Supplies at info@bbmstx.com—and get your storage, fulfillment, and shipping handled by a team that knows medical compliance isn't optional.
Frequently Asked Questions
What is medical distribution?
Medical distribution is the process of warehousing, managing, and delivering medical products—including supplies, devices, and equipment—from manufacturers to end users such as hospitals, clinics, pharmacies, or direct consumers, while maintaining regulatory compliance throughout the supply chain.
What certifications should a medical supply distributor have?
Look for FDA registration/clearance, ISO 13485 compliance (especially for medical devices), GMP standards, and HIPAA-safe data handling practices. Together, these confirm products are stored, packed, and shipped without regulatory risk.
What is the difference between a medical distributor and a 3PL?
A traditional distributor buys and resells medical products at scale, typically to healthcare facilities. A 3PL manages storage, packing, and shipping on behalf of a product company without taking ownership of inventory, which gives smaller and specialized businesses more flexibility and customization.
How do I choose the right medical supply distribution partner for my business?
Start with three filters:
- Verify regulatory credentials (FDA, ISO 13485, GMP)
- Confirm storage and handling infrastructure fits your product requirements
- Assess whether they offer tailored solutions or only standardized enterprise systems
What regulations govern medical supply distribution in the US?
The main frameworks to know:
- FDA: Oversees medical device packaging and handling
- DSCSA: Governs pharmaceutical supply chain security
- GMP: Sets standards for storage and handling practices
- HIPAA: Applies when distribution involves patient-identifiable data
Can small medical product companies use a 3PL for distribution?
Yes. 3PLs are often the right fit for small-to-mid-sized medical product companies. They provide compliant warehousing, custom packing, and shipping without the volume minimums that large enterprise distributors require, making them practical from day one.


