
Introduction
Most fitness and wellness businesses struggle with the same problem: a return policy exists, but the operational process behind it doesn't. Products range from general wellness gear to FDA-regulated devices, and each category carries different handling, compliance, and resale requirements. Getting the process wrong affects more than customer satisfaction — it can expose you to regulatory risk and quietly drain inventory recovery value.
With 19.3% of online sales projected to be returned in 2025, totaling $849.9 billion across retail, the stakes are high. Processing each return costs between $25 and $30, making efficient workflows critical.
This guide walks through the exact steps to handle returns, what factors shape the process, what to prepare in advance, and mistakes to avoid.
TLDR
- Every return needs inspection, classification by product type and reason, and a disposition decision — restock, restore, or dispose
- FDA-regulated wellness devices carry stricter return handling requirements than general wellness products — misclassifying them creates compliance exposure
- Not all returned products can be restocked; condition, contamination risk, and regulatory status determine what happens next
- A documented returns process and compliant infrastructure — built before returns happen — prevents costly, avoidable errors
- Businesses processing high return volumes benefit from working with a compliant 3PL that offers returns processing and product restoration capabilities
How to Handle Returns for Fitness and Wellness Products
A structured, step-by-step approach protects both the business and end consumer, especially when health-related products are involved. The following five steps create a repeatable, compliant process.
Step 1: Receive and Log the Returned Item
Logging every return immediately upon receipt is essential. Capture these details:
- Return reason
- Product SKU
- Order number
- Condition on arrival
- Date received
Physical receiving process:
- Check that the return matches the RMA (Return Merchandise Authorization)
- Verify packaging integrity
- Note any visible damage or contamination
- Photograph condition if damage is present
For higher-volume operations, using a dedicated returns management system or 3PL receiving workflow keeps this process consistent and auditable. Without immediate logging, returns can sit unprocessed, creating inventory discrepancies and delayed refunds.
Step 2: Inspect and Assess Product Condition
Inspection goes beyond surface-level damage checks. For fitness and wellness products, assess:
- Whether the item was used
- If hygienic concerns apply (personal care devices, wearables)
- Whether original components are present
- Functional integrity of moving parts or electronics
Assign condition categories during inspection:
- Unopened/resellable: packaging intact, no signs of use
- Opened but intact: box opened, product unused, all components present
- Functional but used: shows wear, still operates correctly
- Damaged/defective: non-functional or cosmetically impaired
- Non-resellable: contamination risk, missing parts, or safety concern
For FDA-regulated wellness devices, inspection should also flag any product that may require testing or certification before re-entry into inventory.
Step 3: Classify the Return by Reason and Product Type
Classification determines the entire downstream process. Use two axes:
1. Return Reason:
- Defective
- Damaged in shipping
- Wrong item
- Customer dissatisfaction
- Expired
2. Product Type:
- General wellness product (low-risk, lifestyle-focused)
- FDA-regulated medical or wellness device
A damaged general wellness item (yoga mat, foam roller) has different options than a returned blood glucose monitor or TENS unit. The FDA's General Wellness Policy distinguishes between low-risk lifestyle products and cleared medical devices, changing compliance obligations for handling and resale.
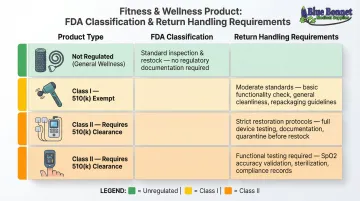
Example Classification Map:
| Product | FDA Class | Return Handling Requirement |
|---|---|---|
| Foam rollers, resistance bands | Not regulated | Standard inspection, restock if undamaged |
| TENS units | Class II | Requires 510(k), stricter restoration protocols |
| Pulse oximeters | Class II | Requires 510(k), functional testing before restock |
| Therapeutic massagers | Class I | 510(k) exempt, moderate handling standards |
Step 4: Determine Product Disposition
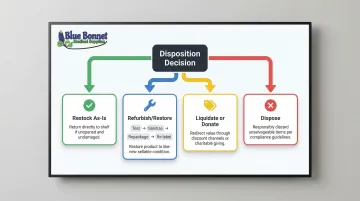
Once classified, choose one of four disposition paths:
Restock as-is: Unopened, undamaged items that meet original specifications go directly back into inventory.
Refurbish or restore: Functional but used or cosmetically imperfect items may be eligible for restoration. This includes:
- Testing functionality
- Sanitizing surfaces
- Repackaging with new materials
- Re-labeling with updated information
This requires FDA and GMP-compliant facility standards. Businesses handling medical-grade fitness products should partner with a 3PL that has documented product testing and restoration capabilities — Bluebonnet Medical Supplies provides this service for returned medical and wellness items.
Liquidate or donate: Functional items that cannot be resold at full price.
Dispose: Defective, contaminated, or non-compliant items.
The FDA distinguishes between "servicing" (returning a device to original specifications) and "remanufacturing" (significantly changing performance, safety, or intended use). Remanufacturing triggers establishment registration, listing, and Quality Management System Regulation (QMSR) compliance.
Step 5: Process the Refund, Update Inventory, and Communicate
Once disposition is determined, trigger the refund or replacement according to your stated return policy:
- Full refund for defective or damaged items
- Partial refund for opened but functional items (if policy allows)
- Exchange for wrong items or customer preference
Inventory updates:
- Restocked items must be logged back into the system with accurate condition data
- Non-resellable items must be written off to prevent phantom inventory
- Restored items should be flagged with restoration date and testing results
Customer communication at each stage:
- Acknowledgment when the return is received
- Confirmation of the refund timeline
- Resolution message once refund is issued
Consistent communication at each stage reduces disputes and creates a documented record — useful if a return escalates to a chargeback or compliance inquiry.
Key Factors That Affect How You Process Returns
The right process for one product or business may not apply to another. These variables determine how complex or straightforward each return becomes.
Factor 1: Product Regulatory Classification
The difference between general wellness products and FDA-regulated wellness devices is critical.
General wellness products (low-risk lifestyle items):
- Fitness trackers (wellness-only claims)
- Foam rollers
- Yoga mats
- Resistance bands
FDA-regulated wellness devices:
- TENS units (Class II, 510(k) required)
- Pulse oximeters (Class II, 510(k) required)
- Blood pressure monitors (Class II, 510(k) required)
FDA-regulated products returned to inventory may need to meet specific documentation and handling standards to remain compliant for resale.
Factor 2: Return Reason
The return reason shapes every subsequent decision:
- Defective units typically require vendor escalation or a formal warranty claim
- Carrier-damaged items trigger a separate claims process with the shipping carrier
- Unopened remorse returns follow the simplest path back to resalable inventory
Documenting return reasons over time also reveals patterns that can reduce future return rates, such as product description mismatches or packaging weaknesses.
Factor 3: Product Condition and Hygiene Risk
Personal-use wellness items carry hygiene considerations that limit restocking options:
- Massage guns
- Wearable fitness tech
- Skin wellness devices
Most used personal care devices can't be resold, even when still functional. Condition assessment must account for contamination risk — particularly for anything that contacts skin directly. That determination drives whether a returned unit gets restored, liquidated, or disposed of.

Factor 4: Return Volume and Operational Scale
Businesses with low return volumes may handle returns manually in-house. Higher volumes require:
- A formalized returns management workflow
- Dedicated receiving area
- Potentially third-party processing
Scale also affects cost. At higher volumes, the cost of inspection, testing, and restoration per unit must be weighed against the recovery value of the restocked item.
What to Prepare Before Processing Returns
Setting up your returns process after problems appear almost always creates inconsistency, compliance gaps, and inventory errors. Getting the infrastructure right before returns arrive saves time and prevents costly mistakes down the line.
Return Policy and RMA System
A clear return policy for fitness and wellness products should include:
- Eligible return window (typically 30 days)
- Acceptable return conditions
- Who covers return shipping
- Refund method (original payment, store credit, exchange)
- Product exclusions (opened hygiene products, used personal care items)
A Return Merchandise Authorization (RMA) system ensures every return is pre-authorized, tracked, and linked to a reason code before it reaches the warehouse. This prevents unauthorized returns and simplifies receiving.
Facility and Compliance Readiness
If restoring or restocking wellness devices, the receiving facility must meet relevant standards. For medical-grade products, this includes:
- FDA-compliant storage conditions
- Proper handling protocols
- Documented procedures for testing and restoration
For businesses without compliant in-house infrastructure, partnering with a certified 3PL ensures returned products are handled without creating liability. Bluebonnet Medical Supplies, for example, integrates FDA, ISO, and GMP compliance into its standard returns and fulfillment workflows — so documentation, labeling, and handling requirements are covered without additional setup on your end.
Common Mistakes When Handling Fitness and Wellness Product Returns
Restocking Returned Products Without Inspection
Even "unopened" returns can arrive damaged, tampered with, or missing components. Reselling them without inspection creates real liability — particularly for health-related items where product integrity directly affects the end user. Verify condition at receiving before anything goes back to shelf.
Treating All Wellness Products the Same
General wellness items and regulated medical devices follow entirely different return rules. What's acceptable for a yoga mat — a visual check and restock — is not acceptable for an FDA-cleared device. That distinction needs to happen at the receiving stage, not after the fact.
Ignoring Return Data
Processing returns without analyzing them is one of the costlier oversights in operations. Patterns in return reasons point directly to fixable problems that keep generating losses:
- Poor packaging leading to transit damage
- Inaccurate product listings causing expectation mismatches
- Fragile components that fail under normal use
Track return reasons, analyze trends quarterly, and address root causes.
Lacking a Documented Disposal Process
Non-resellable items need a formal disposal or liquidation path. Improperly disposing of FDA-regulated devices can trigger compliance violations, and without a documented process, audits become difficult to navigate cleanly.
Frequently Asked Questions
Frequently Asked Questions
How do I return fitness and wellness products?
The process typically starts with contacting the seller or retailer to request an RMA. Package the item securely and ship it back within the stated return window. The seller issues a refund or replacement once they receive and inspect the item.
What reasons qualify for a refund on fitness and wellness products?
Most sellers accept returns for defective or damaged products, wrong items shipped, and in some cases buyer dissatisfaction within a set window. Opened personal care or hygiene items are commonly excluded from refund eligibility.
Can used fitness equipment be restocked and resold after a return?
It depends on the product type, condition, and applicable regulations. Reusable equipment (resistance bands, weights) may be restored and resold if functional, while personal-use devices with hygiene concerns typically cannot be restocked.
How do FDA regulations affect the return of wellness devices?
Products classified as FDA-regulated medical devices (even low-risk wellness devices) must be handled, stored, and potentially tested to meet compliance standards before re-entering inventory. The FDA's General Wellness Policy distinguishes between low-risk lifestyle products and regulated devices.
What should a return policy for health and wellness products include?
A solid return policy covers:
- Return window (typically 30 days)
- Product condition requirements
- Exclusions for opened or hygiene-sensitive items
- Refund method (store credit, original payment, exchange)
- Who covers return shipping costs
When should a business outsource fitness and wellness product returns to a 3PL?
Outsourcing makes sense when return volumes are high, products require regulatory-compliant handling, or your team lacks capacity for proper inspection and restoration. A specialized 3PL — like Bluebonnet Medical Supplies — can manage FDA-compliant handling, product testing, and restoration workflows without you building that infrastructure in-house.


