
Introduction
When a temperature excursion renders a batch of biologics ineffective or a vaccine shipment arrives compromised, the consequences extend far beyond financial loss—patient safety hangs in the balance, and regulatory penalties quickly follow. The FDA actively enforces compliance through warning letters and recalls, while temperature failures cost the pharmaceutical industry an estimated $35 billion annually.
That risk is growing. By 2027, 50% of new medicines entering the market will require cold storage, making packaging vendor selection one of the most consequential supply chain decisions a pharmaceutical company faces.
This guide breaks down the top cold chain packaging companies for pharmaceuticals, what distinguishes each one, and the criteria worth using to evaluate them.
TL;DR
- Cold chain packaging protects temperature-sensitive drugs using insulated shippers, phase change materials, and vacuum insulation panels
- Leading companies include Sonoco ThermoSafe, Pelican BioThermal, Cold Chain Technologies, Softbox Systems, and va-Q-tec
- Evaluate vendors on validated temperature ranges, passive vs. active systems, GDP/ISO/GMP certifications, and reusability
- Compliant downstream storage and fulfillment are just as critical as the packaging itself
- Match packaging validation data to your shipment routes and compliance requirements
Overview of Cold Chain Packaging in the Pharmaceutical Industry
Pharmaceutical cold chain packaging encompasses insulated shippers, gel packs, phase change materials (PCM), and vacuum insulation panels (VIP) designed to maintain specific temperature ranges—such as 2–8°C for refrigerated products, -20°C for frozen items, or -70°C for ultra-cold therapies—during storage and transportation.
The global pharmaceutical cold chain packaging market reached $17.93 billion in 2024 and is projected to hit $63.30 billion by 2033, growing at 15.3% annually. This surge reflects the rising demand for biologics and vaccines—a trend driven largely by the expansion of biologic therapies and mRNA-based vaccines requiring strict thermal controls.

One distinction worth clarifying: packaging manufacturers and logistics providers (carriers and 3PLs) serve different roles. Packaging protects product integrity during transit. A compliant logistics partner handles the storage, handling, and regulatory chain of custody that keeps temperature-sensitive products viable from warehouse to final delivery. Both are essential — and often confused when buyers are evaluating vendors.
Top Cold Chain Packaging Companies for Pharmaceuticals
Not every cold chain packaging vendor is equipped to handle pharmaceutical-grade requirements. The companies below were evaluated on five criteria:
- Temperature performance validation across required ranges
- Packaging format diversity (parcel to pallet scale)
- Regulatory certifications and GDP/ISTA compliance
- Geographic availability and logistics network reach
- Proven pharmaceutical and biopharma client track records
Sonoco ThermoSafe
Sonoco ThermoSafe, a division of Sonoco Products Company, is one of the largest pharmaceutical cold chain packaging manufacturers globally, offering comprehensive solutions from parcel-level insulated shippers to full pallet-scale containers.
Its validated systems cover 2–8°C, 15–25°C, and frozen ranges in both single-use and reusable formats. The Orion r® rental program adds integrated telemetry for real-time tracking with up to 144 hours of thermal protection — useful for extended international lanes.
| Specification | Details |
|---|---|
| Temperature Range | 15–25°C (CRT), 2–8°C (refrigerated), -20°C (frozen), -70°C (ultra-cold) |
| Packaging Types | Envoy® EPS shippers, ChillTech® PCM systems, Orion r® reusable containers, VIP-based pallet shippers |
| Certifications | ISTA-certified testing labs, ASTM standards, EU GDP alignment, PDA Technical Report 39 conformance |
Pelican BioThermal
For operations prioritizing reusable systems and rental logistics, Pelican BioThermal is a strong fit. The company specializes in temperature-controlled containers for clinical trials, biopharma, and commercial pharmaceutical distribution — with a heavy emphasis on rental and leasing models that reduce per-shipment costs.
The Crēdo line of reusable containers uses PCM and VIP technology to deliver up to 168 hours of protection. The Crēdo on Demand™ rental model eliminates the need to manage container inventory — a practical advantage for smaller shippers or variable-volume operations.
| Specification | Details |
|---|---|
| Temperature Range | 15–25°C, 2–8°C, ≤-18°C, ≤-40°C, ≤-60°C (ultra-cold for cell and gene therapies) |
| Packaging Types | Crēdo Cube™ (parcel), Crēdo Vault™ (bulk), CoolGuard single-use, CoolPall pallet shippers |
| Certifications | ISTA 7D qualified, ISO 9001:2015 certified quality management system |
Cold Chain Technologies (CCT)
Cold Chain Technologies is a US-based manufacturer specializing in passive thermal packaging—including gel packs, insulated shippers, and next-generation PCM solutions—serving pharmaceutical, biotech, and clinical diagnostics sectors.
CCT's ReNew program for reusable container management backs up its sustainability positioning with data: life cycle assessments show a 46% reduction in fossil fuel use and 59% decrease in GHG emissions over 20 trips versus single-use EPS. Custom engineering services are also available for non-standard shipment profiles.
| Specification | Details |
|---|---|
| Temperature Range | 15–25°C (CRT), 2–8°C (refrigerated), -15°C to -25°C (frozen), -20°C to -60°C (dry ice) |
| Packaging Types | EcoFlex® Extended (96–144 hour VIP/PCM systems), GTS Excel gel packs, KoolTemp foam shippers, 100% curbside recyclable options |
| Certifications | ISO 9001:2015 certified, ISTA 7D and ASTM testing standards alignment |
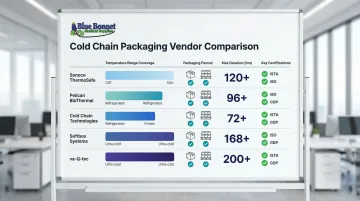
Softbox Systems
Softbox Systems, acquired by CSafe Global in 2021, provides high-performance passive temperature-controlled packaging with a focus on pharmaceutical and clinical supply chains, offering both parcel-level and pallet-level solutions.
The headline differentiator is duration: some configurations reach up to 240 hours of thermal protection using advanced VIP technology. Regulatory alignment with EMA and FDA distribution guidelines comes through the broader CSafe global network, which also supports reverse logistics.
| Specification | Details |
|---|---|
| Temperature Range | +2°C to +8°C, +15°C to +25°C, -25°C to -15°C, <-60°C (ultra-cold) |
| Packaging Types | Softbox VIP reusable parcel shippers (96–240 hour duration), Softbox EPS single-use (72–96 hours), Silverpod systems |
| Certifications | ISTA 7D summer and winter profile qualification, CSafe global network integration for reverse logistics |
va-Q-tec
va-Q-tec is a German-based manufacturer and logistics service provider known for vacuum insulation panel (VIP) technology, offering high-performance passive containers and a rental container network for pharmaceutical shippers.
Its VIP-based containers outperform conventional foam systems significantly, achieving up to 233 hours below -60°C. The TempChain Service Network handles global container rental and management, making va-Q-tec particularly well-suited for ultra-cold applications in mRNA, cell therapy, and gene therapy distribution.
| Specification | Details |
|---|---|
| Temperature Range | +25°C down to -80°C (including ultra-cold ranges for mRNA and CGT) |
| Packaging Types | va-Q-tainer® parcel boxes, va-Q-pal® pallet shippers, va-Q-proof rental containers |
| Certifications | ISO 9001:2015 certified, GDP-compliant transport design, validated to EU and WHO standards |
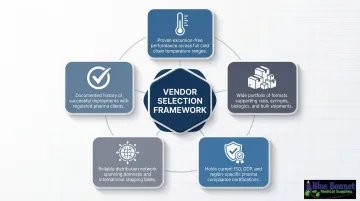
How We Chose the Best Cold Chain Packaging Companies
Not all cold chain packaging vendors are equal — and in pharmaceutical logistics, the wrong choice creates compliance risk, not just operational headaches. We evaluated companies on five primary criteria:
- Validated temperature performance across key pharmaceutical ranges (2–8°C, 15–25°C, frozen, ultra-cold)
- Packaging format diversity from parcel to pallet scale
- Regulatory certifications and testing standards alignment (ISTA, GDP, ISO)
- Geographic availability and supply reliability
- Track record with regulated pharmaceutical clients
Even with strong criteria, buyers often undermine their own decisions. These are the most common mistakes to avoid:
- Choosing based on price alone without checking validation data
- Selecting single-use systems when route duration exceeds tested performance
- Overlooking whether the vendor supports specific distribution regions and regulatory markets
One factor that often goes unaddressed: packaging performance only holds if the surrounding logistics chain is equally compliant. A pharmaceutical product packed correctly can still fail a GDP audit if it moves through non-compliant storage or fulfillment. For businesses that need warehousing and fulfillment to match their packaging standards, working with an FDA, GMP, and ISO-compliant 3PL like Bluebonnet Medical Supplies extends compliance beyond the box — through documented handling, chain of custody, and regulatory-ready storage.
Conclusion
The right cold chain packaging partner directly impacts product efficacy, patient safety, and regulatory compliance. Decisions should be based on validated performance data, certification depth, and fit for specific distribution routes—not just brand recognition.
When evaluating vendors, prioritize these steps:
- Request validation documentation and route-specific performance data from every shortlisted supplier
- Compare reusable vs. single-use cost-per-shipment models against your volume and lane characteristics
- Confirm that your fulfillment partner can maintain temperature integrity through storage and distribution—packaging alone isn't sufficient for end-to-end compliance
If you need a fulfillment partner with FDA, ISO, and GMP compliance to handle storage, packing, and shipping of pharmaceutical or medical products, reach out to Bluebonnet Medical Supplies at info@bbmstx.com to discuss a logistics solution built around your specific needs.
Frequently Asked Questions
What is a pharmaceutical cold chain?
A pharmaceutical cold chain is a continuous sequence of temperature-controlled storage and transport steps that keep a drug product within its required range from manufacture to the patient. Key ranges include 2–8°C for refrigerated products and -20°C or below for frozen products.
What is the difference between active and passive cold chain packaging?
Passive systems use pre-conditioned materials like gel packs or phase change materials to maintain temperature without power, while active systems use powered refrigeration or cooling units. Passive systems are more common for parcel-level pharmaceutical shipments due to simplicity and cost-effectiveness.
What temperature ranges do pharmaceutical cold chain packaging systems cover?
Standard ranges include controlled room temperature (15–25°C), refrigerated (2–8°C), frozen (-20°C), and deep frozen (-70°C or below for advanced therapies). Packaging must be validated specifically for the target range and shipment duration to ensure product integrity.
What certifications should cold chain packaging companies have?
Reputable companies should hold ISTA (International Safe Transit Association) certification for tested configurations, align with GDP (Good Distribution Practice) guidelines, and meet ISO standards. FDA-regulated markets also require packaging performance within labeled conditions per 21 CFR requirements.
Are reusable cold chain packaging systems more cost-effective than single-use?
Reusable systems typically offer lower cost-per-trip at scale but require reverse logistics infrastructure for cleaning, reconditioning, and return. Single-use systems are better suited for lower-volume, international, or one-way shipment lanes where return logistics are impractical.
How long can passive cold chain packaging maintain temperature?
Performance duration varies by system design, insulation material, and ambient conditions, with validated ranges commonly spanning 24 to 120 hours. Advanced VIP/PCM systems can achieve 240+ hours — always verify the packaging's tested duration covers your worst-case transit time for each lane.


