
Introduction
Pharmaceutical logistics operates under a uniquely unforgiving set of constraints. These aren't ordinary shipments. A single temperature excursion can destroy a $500,000 cell therapy, a missing customs document can halt a container at the border for weeks, and a mislabeled package can trigger an FDA refusal.
The industry loses roughly $35 billion annually due to failures in temperature-controlled logistics, with up to 20% of temperature-sensitive products damaged or degraded during distribution. For small-to-mid-sized medical product businesses, the stakes are even higher: one compliance failure can mean lost revenue, a damaged reputation, and — in the worst cases — harm to patients who depend on these products.
Getting this right takes more than good intentions. This guide breaks down the critical components of pharmaceutical logistics — what it is, why it differs from standard supply chain operations, and how to navigate the most common challenges. You'll find practical solutions covering regulatory compliance, cold chain management, packaging standards, and documentation, plus how partnering with a specialized 3PL can take the complexity off your plate so you can focus on growth.
TLDR
- Pharmaceutical logistics covers storage, handling, and transportation of medical products under strict regulatory and environmental conditions — far more complex than standard freight
- Top challenges: maintaining cold chain integrity (costing the industry $35B annually), navigating multi-jurisdictional compliance, ensuring FDA-compliant packaging, and maintaining real-time supply chain visibility
- Solutions: purpose-built storage facilities, IoT-enabled real-time tracking, compliant packaging systems, and outsourcing to a credentialed pharmaceutical 3PL
- Bluebonnet Medical Supplies handles FDA/GMP/ISO compliance, carrier relationships, and customs documentation — so medical product businesses can scale without building that expertise in-house
What Is Pharmaceutical Logistics and Why Does It Matter?
Pharmaceutical logistics covers the end-to-end process of managing, storing, and transporting pharmaceutical and medical products from manufacturer to end destination — hospitals, pharmacies, and consumers — while maintaining product integrity and meeting regulatory standards.
It differs from standard logistics in one critical way: the combination of high product value, strict regulations (FDA, GMP, GDP), and direct patient safety implications makes every step of the supply chain consequential.
The scale of this sector is massive and accelerating. In 2024, the global pharmaceutical logistics market was valued at $99.33 billion and is projected to reach $208.26 billion by 2033, growing at a CAGR of 8.5%. This growth is driven by the rise of temperature-sensitive biologics, stricter traceability mandates, and the expansion of global distribution networks.
Why Pharmaceutical Logistics Is High-Stakes
The financial and patient-safety consequences of logistics failures are severe:
- $35 billion lost annually to preventable cold chain failures across the industry
- 25% of vaccines arrive degraded due to logistics failures
- 7.1% of all pharmaceutical stock is lost somewhere in the supply chain—a $10.3 billion loss—largely due to expiration and obsolescence
- 15% of FDA drug recalls in FY2022 were related to temperature abuse

Numbers like these explain why rigorous compliance and specialized infrastructure aren't optional — and why the challenges covered in this guide carry real consequences for both revenue and patient outcomes.
Key Components of Pharmaceutical Logistics
Pharmaceutical logistics encompasses several interconnected functions, each requiring specialized expertise and infrastructure.
Core Logistics Functions
The primary logistics operations include:
- Temperature-controlled warehousing and storage — Facilities designed with controlled temperature zones, humidity management, and proper racking systems to prevent product damage
- Compliant packaging and labeling — FDA-cleared materials, tamper-evident seals, and accurate labeling that meets regulatory standards for both domestic and international shipments
- Order fulfillment and distribution — Picking, packing, and shipping operations that maintain product integrity and meet delivery timelines
- International shipping with customs documentation — Managing cross-border logistics with proper certificates, traceability records, and country-specific compliance requirements
Inventory Management: The Foundation
Inventory management in pharmaceutical logistics demands precision at three levels: traceability, rotation, and quarantine.
Package-Level Traceability
Both the FDA's Drug Supply Chain Security Act (DSCSA) and the EU's Falsified Medicines Directive (FMD) mandate electronic traceability at the package level. The FDA recommends Electronic Product Code Information Services (EPCIS) standards to maintain interoperable, audit-ready transaction histories. Every package must carry a unique identifier containing product code, serial number, batch number, and expiration date.
FEFO (First Expiry/First Out) Management
FDA 21 CFR 211.150 mandates that "the oldest approved stock of a drug product is distributed first." Poor expiration management can turn millions of dollars of high-value medicine into hazardous waste. WHO GDP guidelines require the same strict stock rotation to ensure products ship within efficacy windows.
Quarantine Systems
WHO GDP requires that "rejected materials and products should be identified and controlled under a quarantine system designed to prevent their use until a final decision is taken on their fate." Without a functioning quarantine process, compromised products risk entering the distribution chain.
Reverse Logistics: The Overlooked Component
Reverse logistics covers the inspection, testing, and restoration of returned pharmaceutical and medical products — and the scale is significant. Annually, more than 120 million units with a product value exceeding $13 billion flow through the reverse distribution process. Unsaleable returns alone account for 1.5% to 2% of sales, totaling $6 billion to $8 billion.
Reverse distribution is strictly regulated:
- DEA regulations (21 CFR Part 1317) require that controlled substances received for destruction must be destroyed within 30 days
- FDA regulations (21 CFR 211.204) mandate that returned drugs be identified, held, and either destroyed or proven to meet appropriate standards through examination and testing
- Products exposed to improper storage conditions cannot be salvaged and returned to the marketplace
Partnering with a provider capable of receiving, inspecting, testing, and restoring returned products — or ensuring compliant destruction when restoration isn't possible — is a non-negotiable part of pharmaceutical logistics.
Top Challenges in Pharmaceutical Logistics
Regulatory Compliance Across Borders
Pharmaceutical businesses face a layered compliance burden: meeting FDA regulations domestically while navigating varying international standards (GDP guidelines, country-specific import/export rules) when shipping globally. Non-compliance can result in shipments held at customs, product seizures, or costly fines.
The FDA's Detention Without Physical Examination (DWPE) Authority: The FDA can place a product on an Import Alert after discovering a violation and then detain future shipments without physically examining them. Import Alert 66-40, for example, allows the FDA to detain drugs from foreign establishments that don't conform to Current Good Manufacturing Practices (CGMP). If a shipment is refused, it must be destroyed or exported under CBP and FDA supervision within 90 days.
Documentation Complexity: Cross-border shipping requires flawless documentation. The EMA's GDP guidelines require that "the required storage conditions for medicinal products should be maintained during transportation within the defined limits as described by the manufacturers." WHO GDP guidelines mandate that distributors "profile and qualify transport routes" and "select the most suitable methods for protecting time- and temperature-sensitive pharmaceutical products."
Missing or incorrect paperwork on any of the following can halt shipments for weeks and trigger regulatory action:
- Certificates of conformity
- Lot traceability records
- Labeling requirements
- Customs and import/export paperwork
Cold Chain Integrity and Temperature Sensitivity
Beyond documentation, the physical handling of products presents an equally demanding challenge. Maintaining temperature control is the most critical operational risk in pharmaceutical logistics. In 2021, cold chain medicines made up $384 billion, or 32% of the total pharmaceutical market (excluding vaccines and COVID-19 therapeutics).
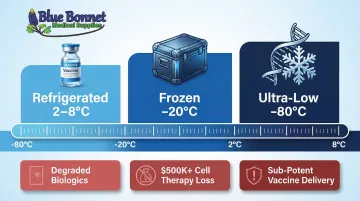
Temperature Requirements Vary Widely:
- Most vaccines require constant refrigeration between 2–8°C
- Some biologics must be stored at -20°C
- Certain mRNA vaccines require ultra-low temperatures (ULT) as low as -80°C
The Consequences of Temperature Excursions: Even brief deviations can cause irreversible damage. A 1–2°C excursion can degrade sensitive biologics, vaccines, or insulin. The financial and safety consequences are severe:
- A single excursion can destroy cell therapies worth $500,000+
- Undetected excursions can deliver sub-potent vaccines to patients, creating direct public health risks
- 15% of FDA drug recalls in FY2022 were related to temperature abuse
Without real-time temperature data during transit, businesses often don't discover a cold chain breach until the product is already compromised. Passive data loggers only reveal the problem after the damage — and the loss — has occurred.
Packaging Compliance and Product Protection
Pharmaceutical packaging must meet FDA standards for materials, labeling accuracy, and tamper evidence. Using non-compliant packaging can result in regulatory rejection, especially for international shipments.
FDA Tamper-Evident Requirements: 21 CFR 211.132 requires that OTC drug products be packaged in a "tamper-evident package"—one having indicators or barriers that provide visible evidence if tampering has occurred.
Prevention of Mix-Ups: 21 CFR 211.130 requires "prevention of mixups and cross-contamination by physical or spatial separation from operations on other drug products" and "examination of packaging and labeling materials for suitability and correctness before packaging operations."
Sourcing FDA-cleared packaging, ensuring correct labeling, and maintaining GMP-compliant packing processes requires expertise and infrastructure that many small-to-mid-sized medical product companies don't have in-house. Specialized 3PLs with existing FDA, ISO, and GMP compliance can close that gap without requiring businesses to build it themselves.
Solutions to Common Pharmaceutical Logistics Challenges
Purpose-Built, Compliant Storage Facilities
The foundation of sound pharmaceutical logistics is a warehouse designed specifically for sensitive medical products. Compliant warehousing is governed by overlapping standards:
FDA 21 CFR 211.142 requires written procedures for warehousing, including:
- Quarantine of drug products before release by the quality control unit
- Storage under appropriate conditions of temperature, humidity, and light
USP <1079> mandates written procedures for qualification of storage and shipping containers, taking into account product category, area layout, and storage volume.
WHO GDP requires that "heating, ventilation and air conditioning systems should be appropriately designed, installed, qualified and maintained" and that "mapping studies for temperature and relative humidity should be done."
Contrast with General-Purpose Warehousing: Storing pharmaceutical or medical products in a facility not built for these requirements is a compliance and quality risk that can result in rejected inventory, regulatory action, and lost revenue. Purpose-built facilities include:
- Controlled temperature zones with validated HVAC systems
- Secured areas with access controls
- Humidity management systems
- Proper racking systems to prevent product damage and enable FEFO rotation
- Quarantine areas for rejected or returned products
End-to-End Supply Chain Visibility and Tracking
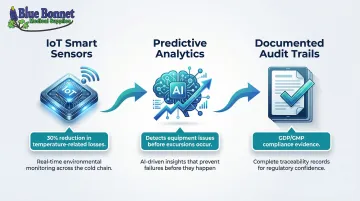
The industry has shifted from passive data loggers to active, IoT-enabled real-time monitoring. Currently, 69% of pharmaceutical companies have adopted automated, real-time cold chain monitoring systems, and 96% say real-time decision-making is essential for supply chain management.
Three capabilities define modern visibility infrastructure:
- IoT smart sensors continuously track temperature, humidity, and location, broadcasting data to cloud dashboards. Organizations using IoT-based monitoring have cut losses on temperature-sensitive products by up to 30%.
- Predictive analytics analyze environmental trends to detect equipment malfunctions and forecast temperature rises—allowing intervention before an excursion occurs.
- Documented audit trails serve as GDP/GMP compliance evidence and resolve disputes with carriers or customers when regulators request proof of proper handling.
Partnering With a Compliant Pharmaceutical 3PL
For small-to-mid-sized medical product businesses, outsourcing to a specialized, compliant 3PL is the most practical solution. The global pharmaceutical 3PL market was valued at $137.25 billion in 2024 and is projected to reach $228.54 billion by 2030, growing at a CAGR of 8.9%. Currently, 60% of manufacturers outsource at least some logistics functions to a 3PL.
The financial case is concrete:
- 10% to 15% savings in transportation costs through shared-resource logistics and carrier relationships
- 20-25% reduction in operational costs by using external partners for commercial operations
- Up to 40% faster speed to market for new products
Compliance Transfer: A credentialed pharmaceutical 3PL maintains FDA clearance, GMP compliance, ISO standards, and HIPAA-safe handling as part of its core operations, transferring that compliance assurance to every client without each client needing to build it independently.
Example: Bluebonnet Medical Supplies is an FDA/GMP/ISO-compliant 3PL built specifically for medical product businesses. Unlike large logistics corporations, Bluebonnet customizes workflows (storage configurations, packing specifications, shipping cadences) to match how each business actually operates.
Bluebonnet also offers returns processing and product restoration services, recovering usable inventory value while keeping products compliant throughout the process.
The Role of a 3PL in Pharmaceutical Logistics
A pharmaceutical 3PL is an outsourced logistics partner that handles warehousing, compliant packing, order fulfillment, carrier coordination, and international shipping. The result: your team focuses on growth while the operational complexity stays off your plate.
The Compliance Advantage
A credentialed pharmaceutical 3PL maintains FDA clearance, GMP compliance, ISO standards, and HIPAA-safe handling as part of its core operations. This means:
- No need to build internal expertise in regulatory frameworks
- Immediate access to validated systems for temperature control, traceability, and documentation
- Reduced risk of compliance failures that can halt shipments or trigger regulatory action
- Audit-ready processes that satisfy FDA, EMA, and WHO GDP requirements

The Flexibility Value
Unlike large, rigid logistics corporations, a boutique pharmaceutical 3PL can customize workflows to match how each business actually operates. This includes:
- Configures storage around your product types and turnover rates
- Tailors packing specs to meet both regulatory requirements and customer expectations
- Aligns shipping cadences with your order patterns and delivery commitments
- Handles custom labeling and kitting for retail, e-commerce, or direct-to-patient distribution
Returns Processing as a Differentiator
Not every 3PL can handle returns — and for medical products, that gap matters. Given that $6 billion to $8 billion in unsaleable returns flow through the reverse distribution process annually, a partner that can receive, inspect, test, and restore returned products while maintaining DEA and FDA compliance directly protects your revenue and reduces write-offs.
Bluebonnet Medical Supplies offers this capability alongside core distribution services. The team checks, tests, and when needed, restores returned items so they can go back on shelves. Few 3PLs offer this level of reverse logistics support for medical products, which makes it a meaningful differentiator for businesses managing high return volumes.
Frequently Asked Questions
What is pharmaceutical logistics?
Pharmaceutical logistics covers the end-to-end management of storing, handling, and transporting pharmaceutical and medical products. The focus is on maintaining product integrity, meeting regulatory requirements (FDA, GMP, GDP), and ensuring timely, safe delivery to hospitals, pharmacies, or consumers.
What types of logistics are used in pharmaceutical logistics?
Pharmaceutical logistics spans several specialized categories:
- Temperature-controlled (cold chain) logistics for biologics and vaccines
- Specialized warehousing with humidity and security controls
- Compliant last-mile delivery
- International freight and customs management
- Reverse logistics for returns and product restoration
What is a 3PL in pharmaceutical logistics?
A 3PL (third-party logistics provider) in pharma is an outsourced partner that handles storage, packing, and shipping of pharmaceutical products on your behalf. These providers hold the regulatory certifications (FDA, GMP, ISO) required to manage medical products compliantly.
What are the biggest challenges in pharmaceutical logistics?
The top challenges include:
- Cold chain integrity — the industry loses an estimated $35 billion annually to temperature failures
- Multi-jurisdictional regulatory compliance across different markets
- FDA-compliant packaging with tamper-evident seals and accurate labeling
- Real-time supply chain visibility for high-value, time-sensitive products
How does temperature control affect pharmaceutical logistics?
Many pharmaceutical products require strict temperature ranges — 2–8°C for vaccines, -20°C for some biologics, -80°C for certain mRNA products. Any deviation during storage or transit can compromise potency, trigger regulatory non-compliance, or cause total product loss, making purpose-built cold storage and real-time monitoring non-negotiable.
How do I ensure regulatory compliance when shipping pharmaceutical products?
Start by working with FDA/GMP-certified partners for storage and packing. From there:
- Maintain accurate documentation and lot traceability using EPCIS standards
- Use compliant packaging with tamper-evident seals and proper labeling
- Verify customs requirements and GDP standards for each destination country before international shipments


