
Introduction
Peak season fulfillment failures represent one of the costliest and most preventable operational challenges for medical product businesses, where predictable demand spikes collide with strict regulatory requirements. A 3% order error rate costs between $705,000 and $1.4 million annually for a warehouse processing 500,000 orders, while drug and medical supply shortages cost U.S. hospitals nearly $360 million per year in additional labor expenses to manage workarounds.
For medical product businesses, the stakes are higher than a delayed shipment. FDA civil monetary penalties reach $35,466 per device violation, meaning a single bad peak season can compound into regulatory and financial damage that outlasts the holiday rush. This guide breaks down the most common fulfillment failure points — from inventory miscounts to carrier capacity crunches — and what to do about each one before volume spikes hit.
TL;DR
- Peak season strains storage, labor, packaging, and shipping operations all at once
- Common failures stem from poor forecasting, insufficient staffing, carrier shortages, and compliance shortcuts
- Medical product businesses face higher stakes — failures trigger both customer service issues and regulatory violations
- Start 8–12 weeks out: lock forecasts, finalize staffing plans, and confirm compliance protocols before the rush hits
- Proactive planning delivers 20–50% forecast accuracy improvements and 15–25% inventory cost reductions
Why Peak Season Fulfillment Fails
Peak season for medical supplies isn't just a holiday problem. Flu season spikes demand for respiratory devices and PPE. Year-end hospital budgets trigger procurement surges. Q4 e-commerce rushes lock up shipping capacity industry-wide.
These surges don't create new problems — they expose the ones already there. Fulfillment failures during peak happen when inventory, labor, carrier access, and compliance gaps are all stressed at once, with no preparation in place to absorb the pressure.
Failure 1: Inaccurate Demand Forecasting
Relying on gut instinct or outdated sales data instead of historical order trends leads to two costly scenarios:
Stockouts: Lost sales, frustrated customers, and permanent damage to recurring B2B relationships. For Amazon sellers, frequent stockouts lower Inventory Performance Index (IPI) scores, damage organic ranking, and cause loss of Buy Box share. Falling below Amazon's IPI threshold of 450 triggers severe cubic-foot storage limits, preventing sellers from sending enough inventory during peak seasons.
Dangerous overstock: Higher storage costs and compliance risk for expiry-sensitive medical items. Medical devices require stability test data under FDA 21 CFR Part 820 to ensure products remain fit for use within labeled expiration dates. Holding excess inventory increases the risk of product degradation and subsequent regulatory failures.
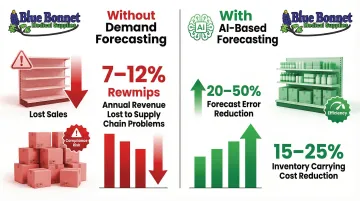
Organizations without demand forecasting tools lose 7–12% of annual revenue to avoidable supply chain problems, while AI-based forecasting reduces errors by 20–50% and cuts inventory carrying costs by 15–25%.
Failure 2: Understaffed and Under-Resourced Operations
Warehouses that don't pre-plan their labor surge create pick-and-pack bottlenecks that slow every order in the system. The transportation and warehousing industry faces chronic labor instability, with annual turnover often exceeding 40%.
The compounding cost of errors:
- A 3% error rate on 500,000 annual orders equals 15,000 mistakes
- Hard costs alone (reshipping, return shipping, re-pick/re-pack labor) bleed $705,000 to $1.4 million
- Average mis-pick costs $30 to $100 per incident to correct
- Best-in-class WERC benchmarks demand ≥99.68% order picking accuracy
Seasonal staff without proper training in FDA labeling requirements or storage protocols mishandle medical products — causing damage, mislabeling, and incorrect packing that creates real compliance exposure at exactly the wrong time.
Failure 3: Carrier and Shipping Capacity Crunches
Carrier capacity doesn't expand to meet demand during peak — it contracts. Pickup frequency drops, surcharges stack up, and businesses without pre-arranged commitments have no leverage to negotiate.
2025/2026 peak surcharges:
- UPS Additional Handling: $10.80 per package
- UPS Large Package: $107.00 per package
- FedEx Additional Handling: $10.90 per package
- FedEx Residential Delivery (Dynamic): Up to $8.75 per package
During peak seasons, the spot-to-contract rate spread collapses to near zero, with spot rates surging 18.9% from mid-November to late December. For medical products specifically, last-minute carrier selection means risking shipment with handlers who don't have the right protocols — a problem that goes well beyond a late delivery.
Failure 4: Compliance Shortcuts Taken Under Pressure
The urgency of a peak season rush causes some businesses to skip or rush through FDA labeling checks, temperature/storage protocol verification, or customs documentation for international shipments.
Critical compliance violations:
- Labeling and UDI failures: Medical devices must bear a Unique Device Identifier (UDI) under 21 CFR Part 801 and 830, containing both device identifier and production identifier (lot/batch, serial number, expiration date). Labeling mix-ups are frequent root causes for Class I and II recalls.
- Import Alert 89-04: The FDA detains products without physical examination (DWPE) if manufacturers violate Good Manufacturing Practices under 21 CFR 820.
- CBP documentation gaps: U.S. Customs requires Country of Origin marking under 19 CFR Part 134 and Affirmation of Compliance (A of C) codes submitted via ACE. Rushed documentation leads to missing codes, triggering immediate customs detentions.

Ship a product outside compliant storage conditions or with missing documentation, and you're looking at customs holds, potential recalls, and damage to your regulatory standing that takes far longer to recover from than the peak season itself.
The Cost of Getting It Wrong
Unaddressed peak season failures create compounding consequences that extend far beyond the immediate crisis.
Financial exposure for your clients:
- FDA civil monetary penalties reach $35,466 per device violation, with aggregate maximums of $2,364,503 in a single proceeding — penalties your clients absorb when fulfillment breaks compliance
- Direct cost of poor quality in medical devices reaches 6.8% to 9.4% of annual sales
- Nonroutine external quality failures (recalls, warning letters, import bans) cost the industry up to $9.5 billion annually
Permanent customer damage:
- Missed revenue windows that can't be recovered
- 68% of B2B buyers report being discouraged by order errors, leading to lost sales and customer attrition
- Hospital contract churn due to supply unreliability — recurring medical supply clients don't return after critical stockouts
Warning Signs You're About to Hit a Peak Season Wall
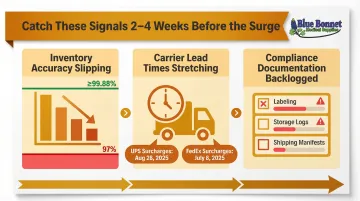
These signals typically appear 2–4 weeks before the surge hits. Catching them early gives you time to adjust before volume makes every problem harder to fix.
- Inventory count accuracy is slipping: Cycle counts are off, reorder triggers aren't firing, and SKU accuracy is declining. Best-in-class accuracy by location is ≥99.88% — dropping into the "Disadvantage" tier (≥90% and <97%) signals systemic issues that worsen as volume climbs.
- Carrier lead times are already stretching: Lengthening delivery windows in pre-peak weeks mean you're heading into a capacity-constrained environment. For 2025/2026, UPS announced Demand Surcharges effective August 28, 2025 (4.4 weeks' notice); FedEx updated surcharges effective July 8, 2025 (11.8 weeks' notice).
- Compliance documentation is backlogged: If labeling, storage logs, or shipping manifests are already falling behind at normal volume, they'll collapse entirely under peak pressure.
How to Prevent Peak Season Fulfillment Failures
Prevention is a pre-season operations checklist — not reactive fixes during the surge, but deliberate decisions made 8–12 weeks in advance that remove the conditions allowing failures to occur.
Prevention Measure 1: Build Demand Forecasts from Real Data
What to do:
- Pull the last 2–3 years of order data segmented by SKU, season, and sales channel
- Layer in new product launches, marketing campaigns, or market trends
- Integrate external signals like flu surveillance data or other epidemiological signals for respiratory seasons, predicting demand spikes weeks in advance
How this blocks the root cause: Accurate forecasts prevent both the stockout scenario (lost sales, damaged IPI scores) and the overstock scenario (excess storage costs, compliance risk for date-sensitive products). In healthcare specifically, service levels should vary by item based on clinical criticality — balancing shelf-life constraints against target fill rates rather than applying a single blanket threshold across all SKUs.
Prevention Measure 2: Plan Staffing and Capacity Before the Season Opens
What to do:
- Identify your peak volume thresholds
- Determine whether current labor, warehouse space, and packing capacity can absorb that volume
- Hire, train, and onboard temporary staff well before the surge
How this blocks the bottleneck: Trained staff — even temporary — make fewer errors with medical products, process orders faster, and reduce the risk of compliance failures caused by mishandling or mislabeling. Document pick-and-pack procedures, compliance handling protocols, and escalation paths so onboarding doesn't require starting from scratch each year.
Prevention Measure 3: Secure Carrier Commitments and Rate Agreements Early
- Negotiate and lock in carrier pickup schedules, rate agreements, and volume commitments at least 8 weeks before expected peak
- Research carriers with medical product handling protocols for regulated shipments
- Diversify fulfillment options by using regional carriers or specialized 3PLs as backup solutions
Pre-committed carrier relationships provide predictable shipping costs, guaranteed pickup windows, and access to medical-compliant options. For international shipments, confirm ocean bookings at least four weeks before sailing dates — blank sailings during peak periods are common and hard to recover from without a confirmed slot.
Prevention Measure 4: Partner with a Compliant 3PL Before Peak Hits
For medical product businesses, working with a 3PL that is already certified for FDA-compliant, ISO, and GMP-aligned fulfillment means compliance is built into the fulfillment process year-round — not something to retrofit under peak pressure.
Mandatory credentials for medical 3PLs:
- FDA Establishment Registration: Facilities involved in production and distribution of medical devices must register annually with the FDA under 21 CFR Part 807
- ISO 13485:2016: The internationally recognized standard for QMS in medical devices, ensuring traceability, contamination control, and risk management
- 21 CFR Part 820 (QMSR): The FDA's Quality Management System Regulation, which now incorporates ISO 13485 by reference

Finding a partner that already holds these credentials eliminates the compliance setup work during your busiest window. Bluebonnet Medical Supplies handles storage, packing, and shipping for medical product businesses — with FDA-cleared packaging services, ISO and GMP compliance, and safe handling procedures built into daily warehouse operations. You scale through peak season without building out internal logistics infrastructure or worrying about a compliance gap under pressure.
When to implement: This isn't a peak-season decision. Onboarding with a compliant 3PL partner ideally happens 2–3 months before peak so inventory is properly transitioned, staff are familiar with your SKUs, and shipping workflows are tested.
Building Fulfillment Resilience Beyond Peak Season
Peak season readiness isn't built in October. The habits and systems you maintain throughout the year determine how well you hold up when volume spikes.
Routine Monitoring
- Establish monthly inventory audits and order accuracy reviews year-round
- Replace annual physical counts with continuous cycle counting
- Count high-velocity "A" items at least once per month, "B" items quarterly, "C" items annually
- Verify that storage conditions, labeling compliance, and documentation standards are consistently met
Training and Process Standards
- Document pick-and-pack procedures, compliance handling protocols, and escalation paths
- Conduct internal audits at planned intervals to ensure your QMS conforms to regulatory requirements
- Maintain rigorous document and record control per ISO 13485 (Clauses 4.2.4 and 4.2.5)
That documentation work pays off twice — it keeps your operation audit-ready year-round and gives your team a clear playbook when peak season pressure hits.
Post-Peak Review
After each peak season, conduct a structured debrief. Systematic After-Action Reviews (AARs) improve team effectiveness and performance by approximately 25% compared to control groups. Document which SKUs stocked out, which carriers underperformed, and where compliance flags appeared — then use those findings to update next year's pre-season plan.
Conclusion
Peak season fulfillment failures are not random. They follow predictable patterns rooted in forecasting gaps, staffing shortfalls, carrier unpreparedness, and compliance pressure. The businesses that weather peak season best aren't the ones that react fastest — they're the ones that prepared months earlier, with the right operational partners already in place.
For medical product businesses, the cost of getting it wrong extends beyond lost sales — a compliance failure during a surge can trigger FDA scrutiny, customs holds, or damaged client relationships that outlast the busy season. Partnering with a 3PL that holds FDA, ISO, and GMP credentials before peak season begins means your regulatory standing stays intact even when order volumes spike. Proactive preparation protects your bottom line and your reputation at the same time.
Frequently Asked Questions
What challenge is common during peak seasons?
The most common challenges include inventory shortfalls, shipping delays, and labor bottlenecks — all of which hit simultaneously when businesses haven't planned capacity in advance. These issues compound when carriers impose surcharges and reduce pickup frequency while warehouse staff struggle with elevated order volumes.
How do you manage logistics planning during peak seasons and supply chain delays?
Start forecasting 8–12 weeks early using historical order data, lock in carrier commitments before surcharges kick in, and coordinate with fulfillment partners ahead of the surge. This lead time allows for staffing adjustments, facility assessments, and contingency planning — rather than scrambling once delays begin.
What is peak season in a warehouse?
Peak season is a predictable window of significantly elevated order volumes — whether tied to holidays, product cycles (flu season for medical supplies), or procurement timelines (year-end hospital budgets) — that places simultaneous stress on storage, labor, and shipping capacity.
How far in advance should medical product businesses prepare for peak season?
Plan for an 8–12 week preparation window. Medical product companies need that lead time to verify compliance documentation, storage readiness, and carrier suitability for regulated shipments — including FDA-compliant labeling and international customs requirements.
What happens to medical product compliance during a peak season rush?
Compliance shortcuts — rushed labeling, skipped storage checks, incomplete customs documentation — are more likely under peak pressure and can result in customs holds, regulatory violations, or product recalls. FDA civil monetary penalties reach $35,466 per device violation, making compliance failures exponentially costlier than the original delay.
How can a 3PL help during peak season fulfillment?
A 3PL with existing warehouse capacity, carrier relationships, and compliance systems absorbs peak volume without requiring last-minute hiring or onboarding. Medical-specialized 3PLs with FDA-compliant, ISO, and GMP-certified operations keep regulatory standards intact throughout the surge — removing the risk of costly shortcuts under pressure.


