
Introduction
Indian medical and health product brands entering the US market face a harsh reality: approximately 25% of medical device import shipments contain documentation errors that trigger FDA holds, and demurrage at major US ports starts at $275 per container per day. Meanwhile, US e-commerce return rates average 20.4%, yet most Indian exporters arrive with no plan for what happens when customers send products back.
Forward logistics—moving products from your Indian facility to US customers—and reverse logistics—managing returns, inspections, and disposition—are two halves of the same operational challenge. Indian brands typically focus exclusively on the outbound journey: export paperwork, freight booking, and customs clearance.
The return journey gets ignored until returned inventory piles up at a US address with no clear process for inspection, restocking, or disposal. That gap is expensive. This guide covers both directions: what it takes to clear US customs cleanly, and what infrastructure you actually need to handle returns without shipping everything back across the ocean.
TL;DR
- Forward logistics covers export documentation in India, international freight (ocean or air), US customs clearance, and last-mile delivery to the customer
- Reverse logistics requires a US-based returns infrastructure — collection, condition inspection, and clear disposition rules (restock, refurbish, or discard) — before you scale
- ISF filing errors trigger $5,000-$10,000 penalties per shipment; HS code misclassification can result in duty reassessments up to 4x the original amount
- Medical device shipments require FDA establishment registration, device listing, and premarket authorization in addition to standard export documentation
- DDP (Delivered Duty Paid) shipping prevents the 10% parcel refusal rate that occurs when customers face surprise customs charges at delivery
What Are Forward and Reverse Logistics?
Forward logistics covers the end-to-end process of moving a product from manufacture in India through international freight, US customs entry, domestic distribution, and final delivery to the customer. It includes procurement, export preparation, carrier selection, documentation filing, duty payment, and last-mile dispatch.
Reverse logistics manages products that flow backward—from the US customer through returns collection, inspection, grading, and disposition (restocking, refurbishment, or disposal). This isn't just "handling returns." It's a complete operational system that determines whether returned inventory recovers value or becomes a write-off.
For Indian brands, forward logistics determines speed and cost of market entry. Reverse logistics determines profitability once you're in market. Medical supplies and devices routinely see return rates of 4-10%, meaning for every 100 units sold, up to 10 will come back. Without a plan, those returns sit idle, lose value, or get abandoned entirely.
How Forward Logistics Works for Indian Brands Selling in the US
The forward logistics chain runs through five stages: export preparation in India → international freight → US customs entry → domestic US distribution → last-mile delivery.
Stage 1: Export Preparation and Documentation in India
Every export shipment requires a core set of documents:
- Commercial invoice — Describes goods, declared value, HS code, and terms of sale
- Packing list — Details package count, dimensions, weight, and contents
- Bill of lading (ocean) or airway bill (air) — Serves as the contract of carriage and receipt of goods
- Export Declaration (Shipping Bill) — Filed with Indian Customs
- IEC (Importer Exporter Code) — Mandatory business identification number issued by India's DGFT for all exports and imports
For medical devices and health products, additional approvals apply. India's CDSCO (Central Drugs Standard Control Organisation) regulates medical device classification (Class A through D) and may require a No Objection Certificate for certain exports. DGFT governs export licensing under India's Foreign Trade Policy.
HS code accuracy matters. US Customs and Border Protection assesses penalties under 19 USC Section 1592 based on culpability:
| Culpability | Penalty | Description |
|---|---|---|
| Negligence | Up to 2x loss of duty or 20% of value | Failure to exercise reasonable care |
| Gross Negligence | Up to 4x loss of duty or 40% of value | Reckless disregard |
| Fraud | Up to 100% of domestic value | Intentional misclassification |

CBP can audit your entire 5-year import history if a pattern emerges. Misclassifying a $50,000 shipment under the wrong tariff code can trigger a $20,000 penalty even if no additional duty was owed.
Stage 2: International Freight — Ocean vs. Air
Transit times and cost structure:
| Mode | Transit Time | Cost Benchmark | Best For |
|---|---|---|---|
| Air Express | 3 days | ~$5/kg (under 150 kg) | Urgent, high-value, low-volume |
| Air Freight | 8-10 days | ~$3/kg (150-500 kg) | Time-sensitive mid-volume |
| Ocean FCL | 30-40 days door-to-door | $1,744-$2,199 per 20ft container | Regular high-volume shipments |
| Ocean LCL | 35-55 days | Per CBM pricing | Volumes between 500 kg and 15 CBM |
Top India-US shipping lanes include Nhava Sheva (JNPT) to New York/Newark, Chennai to Houston, and Mundra to Los Angeles/Long Beach.
ISF 10+2 filing is non-negotiable for ocean freight. The Importer Security Filing must be submitted to US Customs at least 24 hours before cargo is loaded onto the vessel at the Indian port. Late, incomplete, or missing ISF filings trigger liquidated damages of $5,000 per violation, with a $10,000 maximum per shipment. There is no warning system. First violations may be mitigated to $1,000-$2,000, but repeat offenses face full penalties.
ISF is filed by the US importer of record or their customs broker—not the Indian exporter. That said, the exporter must provide accurate data to the broker early enough to meet the 24-hour deadline. If your freight forwarder doesn't manage ISF proactively, your shipment will face holds at the US port.
Stage 3: US Customs Clearance and Last-Mile Delivery
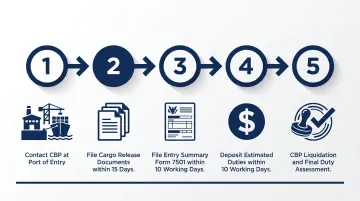
CBP formal entry process:
- Contact CBP at the port of entry to arrange consignment processing
- File cargo release documents within 15 calendar days of arrival
- File Entry Summary (CBP Form 7501) within 10 working days of cargo release
- Deposit estimated duties within 10 working days after entry
- CBP liquidates the entry and finalizes duty assessment
For medical devices, FDA requirements add a parallel layer:
- Valid FDA establishment registration
- Device listing with FDA
- Appropriate premarket authorization: 510(k) clearance, PMA approval, or De Novo classification
- Compliant labeling per FDA requirements
- Adherence to Quality System Regulation (21 CFR 820)
Medical devices do not require Prior Notice filing (that applies only to food and animal feed). Still, a 2013 FDA estimate found that over 25% of medical device import lines contained data errors — improper FDA product codes, inaccurate firm information, or missing registration details. These errors trigger Detention Without Physical Examination, and the problem persists today.
Two active FDA import alerts specifically target Indian manufacturers:
- Alert 89-08 — Authorizes detention of devices lacking approved PMAs or 510(k)s
- Alert 89-04 — Targets manufacturers failing Quality System Regulation compliance
If your device is detained, storage costs accumulate at $50-$150 per day while you resolve the issue. Devices valued at $2,500 or less may face administrative destruction rather than re-export.
US-side warehousing and last-mile:
Indian brands face a choice: self-manage US distribution or partner with a US-based 3PL. Self-management requires establishing a US entity, leasing warehouse space, hiring staff, managing carrier contracts, and maintaining FDA-compliant storage—a significant capital and operational burden.
Partnering with a US 3PL transfers these responsibilities. Bluebonnet Medical Supplies, for example, provides FDA-compliant storage, inventory tracking, order picking and packing, and carrier dispatch — all under ISO and GMP standards. For Indian medical brands, this eliminates the need for a physical US presence while keeping products fully compliant at every step of the chain.
How Reverse Logistics Works for Indian Brands in the US
US e-commerce returns reached $890 billion in 2024, with an average return rate of 20.4%. Health and beauty categories see lower rates (4-10%), but even at 5%, a brand shipping 1,000 units monthly will process 50 returns. Each return costs approximately $40.75 to handle—inspection, restocking labor, carrier fees, and potential disposal.
Indian brands often treat returns as an afterthought. The assumption is that Amazon or the carrier handles it. In reality, unless the brand has its own returns infrastructure, returned goods sit in limbo, lose value, or get abandoned entirely.
Two structural models:
- US-based reverse logistics — Returns are collected, inspected, and processed at a US 3PL facility. Items are restocked into US inventory, refurbished, or disposed of locally.
- Cross-border return to India — Returned items are consolidated and shipped back to India for processing.
Cross-border returns involve repeated customs charges, tariff fluctuations, and multi-country shipping fees. The cost of returning a $30 medical supply item from the US to India often exceeds the product's value. US-based reverse logistics is almost always the economical choice.
Stage 1: Return Initiation and Collection in the US
A return is triggered when a customer requests it (buyer's remorse, wrong item ordered), when a product arrives defective, or during a regulatory recall. The collection process requires:
- Pre-paid return labels issued to the customer or sent via email
- Carrier pickup or customer drop-off at a designated location
- Receiving at a US returns facility with intake scanning and tracking
Without a US partner, Indian brands have no address to print on the return label and no facility to receive returned goods.
Stage 2: Inspection, Grading, and Disposition
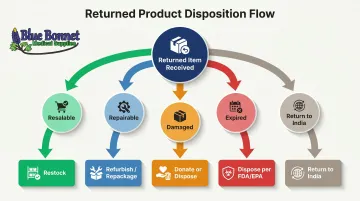
Once goods arrive at a US facility, each item goes through condition assessment before any disposition decision is made:
- Resalable — Unopened, undamaged, within shelf life
- Repairable/Repackagable — Opened but functional; can be repackaged
- Damaged — Cosmetic or functional damage; cannot be resold as-is
- Expired — Past shelf life or regulatory expiration date
Disposition paths:
- Restock — Return to active inventory for resale
- Refurbish/Repackage — Restore to sellable condition through testing and repackaging
- Donate — Send to charitable organizations (tax benefit)
- Dispose — Destroy per FDA/EPA requirements
- Return to India — Consolidate for cross-border return (rare for medical products)
For medical devices, every step of this process must meet FDA and GMP standards. Bluebonnet Medical Supplies handles product testing and restoration in-house — inspecting returned items for functionality, repackaging under FDA-compliant processes, and returning them to active inventory rather than writing them off.
Stage 3: Restocking or Final Disposal
After disposition decisions:
- Items graded as resalable or restored return to active inventory with updated tracking records
- Unsaleable medical items are destroyed with documented disposal records for regulatory and financial compliance
- If cross-border return is chosen, items are consolidated and prepared with full re-import documentation: commercial invoice, packing list, and Indian customs clearance paperwork
Re-importing into India also triggers customs duty reassessment — one more reason most medical product brands keep their returns process entirely within the US.
Key Factors That Affect India-US Logistics Operations
Several variables determine whether an India-US logistics operation runs smoothly or stalls at the border. Getting these right from the start saves time, money, and customs headaches.
Regulatory Compliance
US Customs and Border Protection (CBP) sets strict requirements for documentation accuracy and ISF (Importer Security Filing) submissions. For medical devices specifically, FDA import rules add another layer: product registration, device listing, and premarket authorization must all be in order before goods arrive. Ocean and air shipments also require Automated Manifest System (AMS) filings, which must be submitted within carrier-specific deadlines.
Documentation Accuracy
HS codes, declared values, and product descriptions must match exactly across all documents. Mismatches are the single most common cause of customs holds — and for FDA-regulated products, inaccurate product codes contribute to an industry-wide error rate of around 25%. Double-checking these details before shipment departs India is far less costly than resolving a hold at the port.
Seasonality and Port Congestion
Peak import volumes in Q3 and Q4 put serious pressure on West Coast ports. In September 2024, rail dwell times at LA/Long Beach spiked to 9.25 days — roughly double the pre-surge average. Post-holiday periods (January through February) then bring return volume surges. East Coast ports like NY/Newark and Savannah offer a practical alternative when West Coast congestion is high.
Mode and Route Selection
- Ocean FCL is cost-effective for regular shipments above 15 CBM
- Air freight works for urgent or high-value, low-volume cargo
- Port selection affects both transit time and congestion exposure
US-Side Infrastructure
Brands with reliable US warehousing and 3PL partnerships fulfill orders faster and process returns more efficiently. Without that infrastructure in place, delays compound — and compliance risks rise at every handoff.
Common Challenges and Misconceptions
Misconception: Once goods leave India, the brand's responsibility ends.
In reality, the Indian brand retains responsibility for US customs compliance, duty payment, and delivery unless contractual arrangements (Incoterms) specify otherwise. Under DDP (Delivered Duty Paid), the seller handles all costs and risks through final delivery. Under DAP (Delivered at Place), the buyer pays import duties and taxes.
The cost of getting this wrong adds up fast. For DAP/DDU shipments:
- ~10% of parcels are refused when customers face surprise customs charges at delivery
- Each refusal costs the seller $30–$50 or more in return freight and lost inventory
- One refused delivery erases the duty savings from 5–15 successful DDP shipments
- DDP prevents 48% of cart abandonments caused by unexpected checkout costs

For D2C brands, DDP is the preferred model. The customer pays one price at checkout — no surprise charges, no refusals. That same predictability matters on the reverse side, too.
Misconception: Returns are handled by Amazon or the carrier.
Amazon FBA handles returns for products sold through Amazon, but brands selling through their own websites or other channels need their own returns processing infrastructure. Without it, returned goods accumulate with no clear disposition path.
Misconception: The cheapest shipping option saves money.
For compliance-sensitive goods like medical devices, the lowest freight rate often leads to the highest total cost. A shipment delayed 5 days at LA/Long Beach accrues $1,375 in demurrage charges ($275/day). Factor in an FDA review hold at $100/day, and a 7-day examination adds another $700 before the goods move an inch.
Carriers and brokers with specific medical device experience — Prior Notice familiarity, established FDA relationships — consistently avoid these holds. That expertise is worth far more than the rate difference.
Frequently Asked Questions
What is the shipping cost from India to the USA per kg?
Air express costs approximately $5/kg for shipments under 150 kg; standard air freight runs around $3/kg for 150-500 kg shipments. Ocean freight (LCL) becomes the most economical option above 500 kg and is priced per cubic meter rather than per kg.
How much does a 20-foot container cost to ship from India to the USA?
A 20-foot FCL container from JNPT to Los Angeles ranges from $1,744 to $2,199 depending on season, carrier, and capacity. Rates fluctuate significantly based on port congestion, fuel surcharges, and geopolitical factors.
What is the best way to ship from India to the USA?
There is no single best method. Air freight suits urgent or high-value low-volume shipments; ocean FCL or LCL works better for regular high-volume consignments. For regulated medical products, mode selection must also account for compliance handling and the risk of customs holds.
What documents are required to ship products from India to the USA?
Core documents include a commercial invoice, packing list, bill of lading or airway bill, ISF filing (ocean freight), and any product-specific permits. Medical devices also require FDA establishment registration, device listing, and premarket authorization; electronics with RF components require FCC certification.
How do Indian brands handle product returns from US customers?
The most practical model is US-based returns processing through a 3PL. This avoids expensive cross-border return shipping, allows faster restocking, and—for regulated categories—ensures compliant inspection and disposition without sending items back to India.
What is the difference between forward and reverse logistics?
Forward logistics is the flow of products from manufacturer to end customer; reverse logistics is the return flow after a sale or end-of-use. Indian brands selling in the US need to plan both directions—neglecting reverse logistics leads to operational gaps and avoidable financial losses.


