
Introduction
Medical packaging has a conflict built into it: single-use plastics protect product sterility, yet they account for 2.1 million tonnes of plastic waste generated annually by healthcare systems in the US and Europe. For medical supply businesses, that tension is no longer easy to ignore.
Unlike consumer packaging, medical packaging can't simply be swapped for kraft paper. Regulatory compliance, sterility, and product integrity must come first. FDA standards, ISO 11607 sterile barrier requirements, and GMP protocols create hard constraints that sustainability initiatives must respect. The real question is how to pursue sustainability strategically without compromising safety or compliance.
This article outlines five practical approaches for medical device manufacturers and online medical supply retailers to reduce their packaging footprint while staying fully compliant.
TLDR
- Recyclable mono-material formats and bioplastics reduce plastic waste without sacrificing performance
- Right-sized packaging cuts material use, shipping costs, and carbon footprint simultaneously
- Sustainable sterile packaging is advancing, with recyclable films now meeting ISO 11607 compliance requirements
- Eco-friendly protective packing (paper cushioning, molded pulp) offers quick, low-risk wins
- True sustainability means full life cycle thinking, from raw materials to end-of-life disposal
Why Sustainable Medical Packaging Is No Longer Optional
The numbers are stark. Healthcare plastic waste generates 9.3 million tonnes of CO2e emissions annually, with North America responsible for 1.2 million metric tons. But the pressure to change isn't just environmental—it's commercial and regulatory.
68% of hospitals now have sustainable procurement policies that influence purchasing decisions, and 73% actively purchased environmentally preferable medical supplies in the past year. For medical device manufacturers and distributors, sustainability credentials are increasingly becoming table stakes for winning hospital contracts.
Regulatory bodies are also tightening expectations. Key rules to know:
- EU PPWR: All packaging must be recyclable by 2030; "contact-sensitive" medical device packaging gets an exemption until 2035
- US EPR laws: California, Oregon, Colorado, and Minnesota now require recyclable materials—though FDA-regulated medical devices remain exempt from EPR fees
The catch: medical packaging must still maintain sterility, protect sensitive devices, and meet ISO and FDA standards. Sustainability can't come at the cost of compliance—and that constraint defines the five approaches covered below.
Way 1: Switch to Recyclable or Biodegradable Materials
What's Wrong with Traditional Medical Packaging Plastics
Conventional plastics dominate medical packaging for good reasons: durability, airtight seals, and moldability. But that same durability creates an environmental liability. Most medical plastics are multilayer or composite materials that can't enter standard recycling streams because components can't be separated before processing.
Regulatory and EPR trends in Europe and the US are pushing manufacturers toward recyclable mono-materials. While FDA-regulated devices remain exempt from current EPR mandates, secondary and tertiary packaging (outer shipping cartons and palletizing) used by distributors faces growing compliance costs.
Recyclable Material Options
Mono-material packaging uses a single plastic type—polyethylene (PE) or polypropylene (PP)—throughout the entire package, simplifying recycling since components don't need separation. Conventional multilayer blister packs combine multiple incompatible plastics, making that separation impossible at most recycling facilities.
Leading recyclable options include:
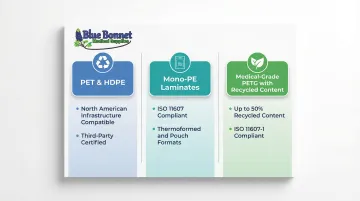
- PET and HDPE: Compatible with existing North American recycling infrastructure and often carry third-party recyclability certification
- Mono-PE laminates: Amcor's Recycle-Ready Medical Laminates are designed for 3D thermoformed packages and 2D pouches while meeting ISO 11607 sterile barrier requirements
- Medical-grade PETG with recycled content: Klöckner Pentaplast's SecondLife® MD incorporates up to 50% certified recycled content while maintaining ISO 11607-1 compliance
Biodegradable and Bioplastic Alternatives
Leading bioplastic materials being explored for medical packaging include:
- Starch-based bioplastics (corn, sugarcane): Plant-derived with controlled lifespans, offering non-petroleum alternatives
- Cellulose-based materials: Can withstand 134°C steam autoclaving where synthetic polymers may deform
- PLA (polylactic acid): Bio-based polymer with established certification pathways
One important caveat on PLA: It requires industrial composting facilities to break down properly — sustained high temperatures and controlled humidity that standard landfills don't provide. In typical landfill conditions, PLA degrades very slowly, often taking months to years.
PLA also suffers severe degradation under gamma irradiation and steam autoclaving, limiting its use primarily to Ethylene Oxide (EtO) sterilization.
Evaluate actual end-of-life infrastructure when choosing materials — not just marketing claims.
Way 2: Embrace Minimalist and Right-Sized Packaging Design
Minimalist packaging design strips away what isn't needed—excess layers, decorative components, oversized containers—leaving only what's structurally and hygienically required. For medical product businesses, this isn't just an aesthetic choice. It directly affects cost, carbon footprint, and shipping efficiency.
Right-sizing means packaging that precisely fits the device or product. This approach:
- Reduces void fill and material waste
- Lowers dimensional weight during shipping
- Cuts freight costs directly
- Decreases carbon footprint per unit delivered
Real-world results:
- Plexus helped a medical device manufacturer replace single-use plastic bags with robust, recyclable tray-based packaging, improving packing density and achieving cost reductions of nearly 50%
- OIA Global's packaging optimization for an endoscopic imaging manufacturer delivered 20% inbound logistics savings and 30% outbound savings
Smaller, lighter packages mean more units per shipment, fewer trucks, and lower emissions per unit. That's a compelling argument for any medical product business balancing sustainability goals with tight margins.
Compliance checkpoint: Minimalist design still has hard floors it can't go below. Key requirements include:
- ISO 11607: Materials must maintain tensile strength, tear resistance, and microbial barrier properties
- FDA 21 CFR 801: Unique Device Identifiers (UDI) must appear in plain-text and barcode formats, with font sizes no smaller than 10 points
- These labeling rules set the absolute minimum dimensions for primary packaging
Run a packaging audit with a compliance-aware partner before implementation to confirm your minimalist redesign clears both standards.
Way 3: Build Sustainability into Sterile and Compliant Packaging
Sterile packaging presents the hardest sustainability challenge in medical packaging. ISO standards require sealed sterile barriers that are waterproof, non-reactive, and uncompromised through storage and transport. This is why single-use plastics dominate, and why biohazard waste rules in surgical settings often prevent packaging from entering recycling streams at all.
Sustainable Sterile Packaging in Practice
Emerging solutions that meet both sterility and sustainability requirements include:
Three commercially available products currently balance compliance with reduced environmental impact:
| Product | Compliance & Application | Sustainability Feature |
|---|---|---|
| Amcor Recycle-Ready Medical Laminates | Designed for medical applications; suitable for 3D thermoformed packages and 2D pouches | Mono-material PE laminate recyclable within mono-PE streams |
| Klöckner Pentaplast SecondLife® MD | ISO 11607-1 and ISO 10993-5 compliant; full sterilization options | Medical-grade PETG with up to 50% certified recycled content |
| DuPont Tyvek 1073B | ISO 11607 compliant; compatible with EO, gamma, e-beam, and controlled steam sterilization | 100% HDPE, certified 100% recyclable at facilities accepting flexible HDPE |

Starch-based and chitosan-based polymers are also advancing through clinical research as an emerging bioplastic category. These materials can create antimicrobial or biodegradable sterile barriers without petroleum-based inputs, though not all currently qualify for sterile applications.
The Operational Challenge
Even when recyclable sterile packaging exists, recycling requires workflow changes. A 2021 pilot program targeting Tyvek and mixed polyolefins successfully diverted 13,792 pounds of plastic waste from landfills over nine months. However, CDC and OSHA Bloodborne Pathogen regulations prevent opened sterile packaging from entering standard recycling streams if potentially contaminated. Recycling must focus strictly on "pre-case" collection—gathering packaging before the patient enters the operating room.
Achieving sustainability in sterile packaging requires both the right materials and the right facility protocols. Choosing recyclable options only delivers results when your handling and collection workflows support them.
For medical product businesses, working with an FDA-cleared, ISO and GMP-compliant 3PL like Bluebonnet Medical Supplies means sustainable packaging choices are carried out within a framework that already meets regulatory requirements — so you're not trading compliance for sustainability.
Way 4: Replace Protective Packing Materials with Eco-Friendly Alternatives
The Problem with Traditional Protective Packing
Polystyrene foam (EPS) is widely used to cushion fragile medical devices during transport, but it contributes heavily to landfill waste and microplastic contamination. Only 3.6% of EPS containers are recycled in the US, and its extremely lightweight structure causes it to fragment rapidly, generating millions of micro- and nanoparticles. As a result, EPS faces growing legislative bans across US states and the EU.
Sustainable Swap Options
Proven alternatives that pass rigorous International Safe Transit Association (ISTA) drop tests include:
- Kraft paper cushioning (such as Ranpak's PadPak) replaced plastic airbags for NeoPharm, cutting transit damage to just 1-2 packages per 100 while reducing labor costs
- Shredded recycled cardboard cushions effectively and decomposes cleanly after delivery
- Starch-based foam peanuts dissolve in water and are certified compostable — no landfill contribution
- Molded pulp inserts suit medical trays and equipment packaging well, offering strong cushioning with full biodegradability

Each of these swaps has cleared rigorous ISTA drop testing, so performance isn't sacrificed for sustainability. That matters most when you're deciding where to start.
The Circular Opportunity
Protective packing represents one of the lowest-risk areas for sustainability upgrades. Because it generally doesn't contact the medical device directly and is unlikely to become biohazard-contaminated, companies can pilot eco-friendly packing for device shipments without touching sterility compliance at all.
Way 5: Think Full Life Cycle Sustainability
Choosing a material labeled "biodegradable" or "recyclable" doesn't automatically make it sustainable. The full picture includes raw material sourcing, manufacturing energy use, transportation emissions, and end-of-life disposal infrastructure. This is where Life Cycle Assessment (LCA) methodology, governed by ISO 14040 and ISO 14044 frameworks, becomes essential.
PLA as a Cautionary Example
PLA plastics were marketed as compostable and carbon-neutral, but they require industrial composting facilities to break down. Without access to those facilities, PLA behaves like conventional plastic in landfills. This illustrates why businesses must audit the real-world end-of-life pathway for any material they choose, not just the label.
A 2021 LCA study compared single-use polypropylene sterilization wrap against reusable Rigid Sterilization Containers (RSCs). While RSCs had an 85% lower carbon footprint, they required a break-even point of 98 use cycles to offset higher initial manufacturing and washing impacts. The lesson: sustainability benefits aren't automatic. They depend on real-world usage patterns.
Logistics as Part of the Sustainability Equation
Packaging doesn't end at the box. How products are stored, packed, and shipped contributes to or detracts from the total carbon footprint. The Global Logistics Emissions Council (GLEC) Framework calculates transport emissions in "tonne-kilometers" (tkm), meaning one tonne of cargo moving one kilometer.
Weight and distance drive the calculation, which makes packaging dimensions and cube utilization critical. Inefficient packaging that ships "empty air" forces distributors to move the same volume of product across more vehicles, directly increasing Scope 3 transport emissions.
Three logistics factors determine how much of that emissions burden your packaging creates:
- Package dimensions: Right-sized packaging eliminates dead space and reduces vehicle trips
- Cube utilization: Maximizing how product fills each shipment lowers the tkm per unit
- Carrier optimization: Consolidating shipments with compliant carriers cuts redundant transport legs

Working with a 3PL partner that applies these principles — like Bluebonnet's FDA/GMP-compliant warehouse operations — ensures sustainability thinking extends beyond the material label and into every shipment your products make.
Frequently Asked Questions
What makes medical packaging different from regular sustainable packaging?
Medical packaging must meet ISO sterile barrier requirements and FDA regulations, meaning sustainability swaps can't compromise product sterility, tamper evidence, or labeling standards. Unlike consumer packaging, medical packaging faces hard regulatory constraints that limit material choices.
Can biodegradable materials meet FDA and ISO sterility standards?
Some bioplastic materials—starch-based and cellulose-based—are being developed and tested for sterile applications, and select products are earning certifications. Not all biodegradable materials currently qualify, though; any candidate material must pass ISO 11607 validation testing before use in a sterile barrier application.
Does sustainable medical packaging cost more than traditional options?
Upfront material costs for bioplastics or certified recyclable materials can be higher. Right-sizing and minimalist design frequently offset that premium through lower material volume, reduced dimensional shipping weight, and fewer returns from damage.
What US regulations govern sustainable medical packaging?
The FDA regulates medical device packaging under 21 CFR 801 and recognizes ISO 11607 for sterility and labeling standards. No single federal mandate currently requires sustainable packaging, though Extended Producer Responsibility (EPR) legislation is advancing at the state level and EU MDR packaging provisions are influencing US importers and manufacturers.
What is the biggest challenge in achieving sustainable medical packaging?
Maintaining a certified sterile barrier while switching to recyclable or biodegradable materials is the central challenge. Most sustainable alternatives haven't matched conventional plastic's sealing and inertness properties at scale, and biohazard waste regulations block post-procedure recycling in surgical settings.
How can small or mid-size medical supply businesses start implementing sustainable packaging?
Start with the lowest-risk wins: swap protective packing materials first (no compliance impact), then right-size packaging dimensions, and then evaluate recyclable outer carton options before tackling sterile barrier materials. A phased approach reduces cost and compliance risk.


